| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1344848 | 980147 | 2009 | 6 صفحه PDF | دانلود رایگان |
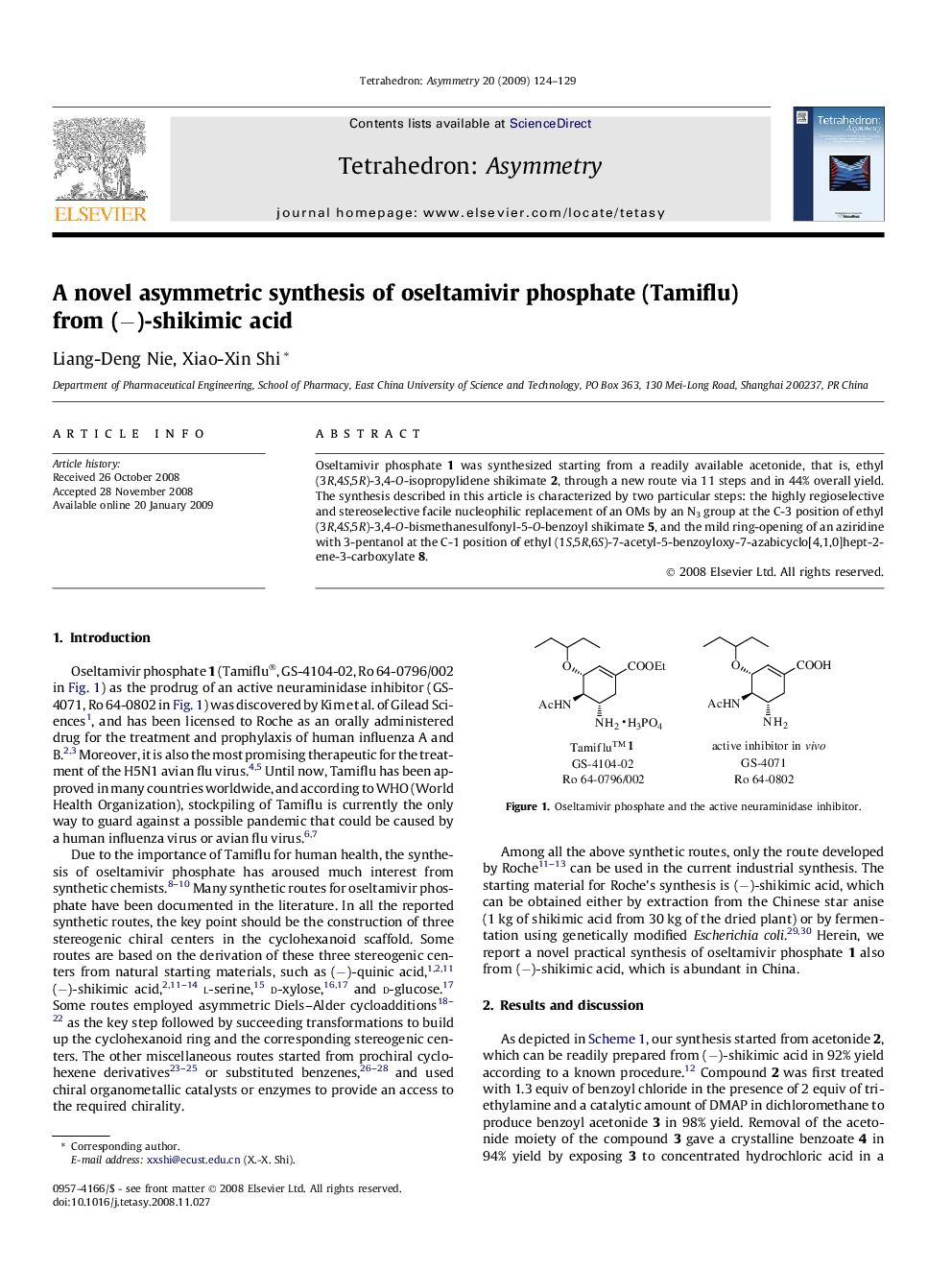
Oseltamivir phosphate 1 was synthesized starting from a readily available acetonide, that is, ethyl (3R,4S,5R)-3,4-O-isopropylidene shikimate 2, through a new route via 11 steps and in 44% overall yield. The synthesis described in this article is characterized by two particular steps: the highly regioselective and stereoselective facile nucleophilic replacement of an OMs by an N3 group at the C-3 position of ethyl (3R,4S,5R)-3,4-O-bismethanesulfonyl-5-O-benzoyl shikimate 5, and the mild ring-opening of an aziridine with 3-pentanol at the C-1 position of ethyl (1S,5R,6S)-7-acetyl-5-benzoyloxy-7-azabicyclo[4,1,0]hept-2-ene-3-carboxylate 8.
Figure optionsDownload as PowerPoint slide
Ethyl (3R,4R,5R)-4-N-acetylamino-3-(1-ethyl-propoxy)-5-hydroxy-cyclohex-1-ene-1-carboxylateC16H27NO5[α]D25=-104 (c 3.0, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4R,5R)
Ethyl (3R,4S,5R)-4-N-acetylamino-3-(1-ethyl-propoxy)-5-methansulfulonyloxy-cyclohex-1-ene-1-carboxylateC17H29NO7S[α]D25=-85 (c 0.7, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4S,5R)
Ethyl (3R,4R,5S)-4-N-acetylamino-5-azido-3-(1-ethyl-propoxy)-cyclohex-1-ene-1-carboxylateC16H26N4O4[α]D20=-44 (c 1.5, CHCl3)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4R,5S)
Osetamivir phosphate (Tamiflu)C16H31N2O8P[α]D20=-39 (c 1, H2O)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4R,5S)
Ethyl (3R,4S,5R)-3,4-O-isopropylidene-shikimateC12H18O5[α]D20=-31 (c 3.0, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4S,5R)
Ethyl (3R,4S,5R)-5-O-benzoyl-3,4-O-isopropylidene-shikimateC19H22O6[α]D25=-55 (c 3.4, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4S,5R)
Ethyl (3R,4R,5R)-5-O-benzoyl-shikimateC16H18O6[α]D25=-122 (c 2.7, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4R,5R)
Ethyl (3R,4S,5R)-5-O-benzoyl-3,4-O-bismethansulfonyl-shikimateC18H22O10S2[α]D25=-135 (c 1.9, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4S,5R)
Ethyl (3S,4R,5R)-3-azido-5-benzoyloxy-4-methanesulfonyloxy-cyclohex-1-ene-1-carboxylateC17H19N3O7S[α]D25=-27 (c 2.8, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3S,4R,5R)
Ethyl (1S,5R,6S)-5-benzoyloxy-7-aza-bicyclo[4,1,0]hept-2-ene-3 -carboxylateC16H17NO4[α]D25=-63 (c 0.8, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (1S,5R,6S)
Ethyl (1S,5R,6S)-7-acetyl-5-benzoyloxy-7-aza-bicyclo[4,1,0]hept-2-ene-3 -carboxylateC18H19NO5[α]D25=-41 (c 1.6, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (1S,5R,6S)
Ethyl (3R,4R,5R)-4-N-acetylamino-5-benzoyloxy-3-(1-ethyl-propoxy)-cyclohex-1-ene-1-carboxylateC23H31NO6[α]D25=-113 (c 1.6, EtOAc)Source of chirality: (−)-Shikimic acidAbsolute configuration: (3R,4R,5R)
Journal: Tetrahedron: Asymmetry - Volume 20, Issue 1, 30 January 2009, Pages 124–129