| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1346109 | 980240 | 2005 | 11 صفحه PDF | دانلود رایگان |
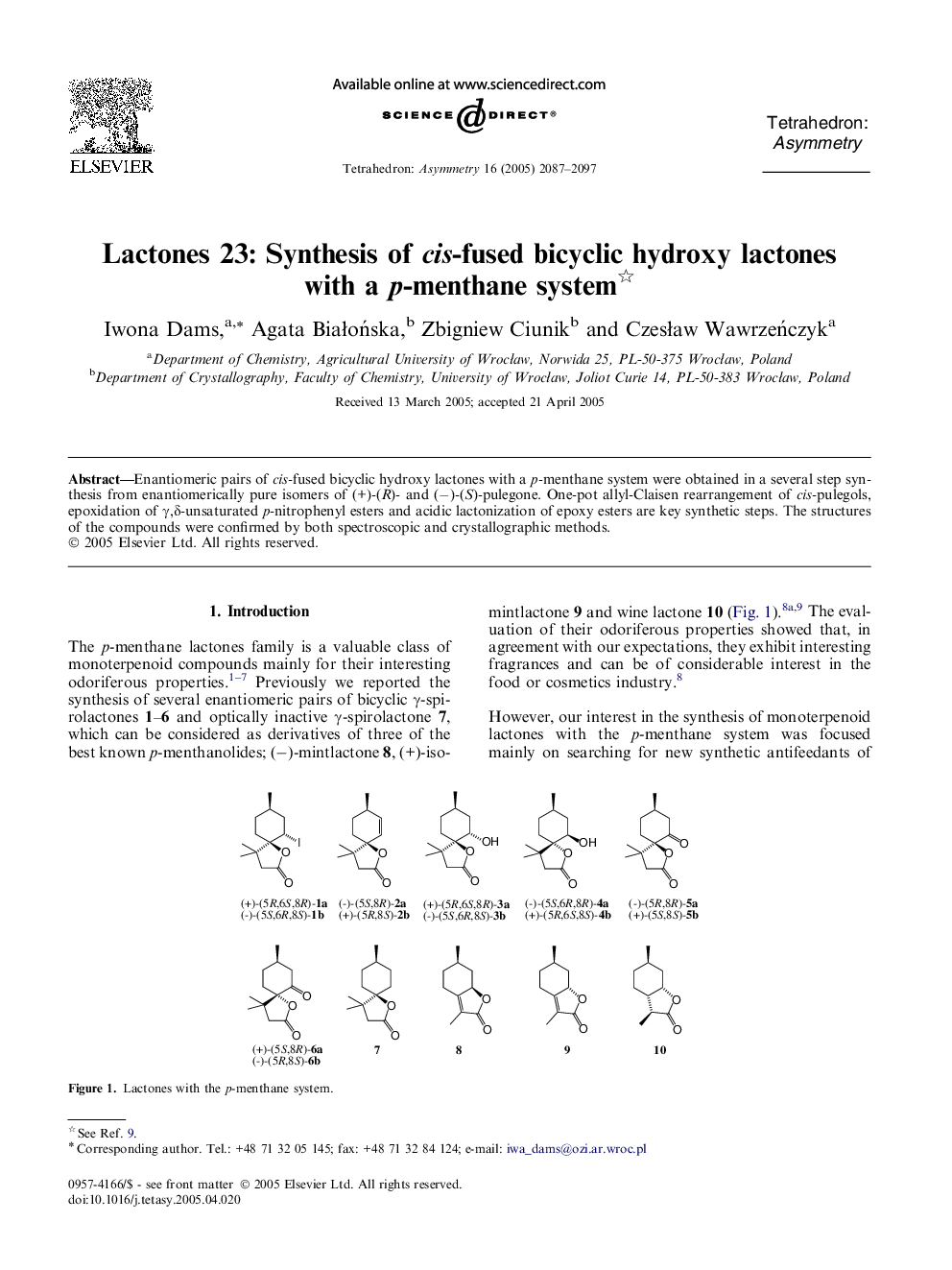
Enantiomeric pairs of cis-fused bicyclic hydroxy lactones with a p-menthane system were obtained in a several step synthesis from enantiomerically pure isomers of (+)-(R)- and (−)-(S)-pulegone. One-pot allyl-Claisen rearrangement of cis-pulegols, epoxidation of γ,δ-unsaturated p-nitrophenyl esters and acidic lactonization of epoxy esters are key synthetic steps. The structures of the compounds were confirmed by both spectroscopic and crystallographic methods.
Figure optionsDownload as PowerPoint slide
Ethyl (2′-isopropyliden-5′-methylcyclohex-1′-yl)acetateC14H24O2Ee = 100%[α]D25=+30.5 (c 3.16, acetone)Absolute configuration:(1′R,5′R)
p-Nitrophenyl (2′-isoporopyliden-5′-methylcyclohex-1′-yl)acetateC18H23NO4Ee = 100%[α]D25=+14.3 (c 3.45, acetone)Absolute configuration:(1′R,5′R)
p-Nitrophenyl (2′-isopropyliden-5′-methylcyclohex-1′-yl)acetateC18H23NO4Ee = 100%[α]D25=-73.9 (c 1.29, acetone)Absolute configuration:(1′S,5′R)
p-Nitrophenyl (2′,7′-epoxy-2′-isoporopyl-5′-methylcyclohex-1′-yl)acetateC18H23NO4Ee = 100%[α]D25=+37.5 (c 3.11, acetone)Absolute configuration:(1′R,2′R,5′R)
1-Hydroxy-2,2,8-trimethyl-3-oxabicyclo[4.4.0]decan-4-oneC12H20O3Ee = 100%[α]D25=+43.8 (c 1.46, acetone)Absolute configuration:(1S,6R,8R)
1-(1′-Hydroxy-1′-methylethyl)-4-methyl-9-oxabicyclo[4.3.0]nonan-8-oneC12H20O3Ee = 100%[α]D25=-21.0 (c 1.52, acetone)Absolute configuration:(1R,4R,6R)
p-Nitrophenyl (2′,7′-epoxy-2′-isoporopyl-5′-methylcyclohex-1′- yl)acetateC18H23NO4Ee = 100%[α]D27=-44.1 (c 1.42, acetone)Absolute configuration: (1′S,2′S,5′R)
p-Nitrophenyl (2′,7′-epoxy-2′-isoporopyl-5′-methylcyclohex-1′-yl)acetateC18H23NO4Ee = 100%[α]D27=-14.3 (c 0.97, acetone)Absolute configuration: (1′S,2′R,5′R)
Journal: Tetrahedron: Asymmetry - Volume 16, Issue 12, 20 June 2005, Pages 2087–2097