| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1346192 | 980247 | 2013 | 14 صفحه PDF | دانلود رایگان |
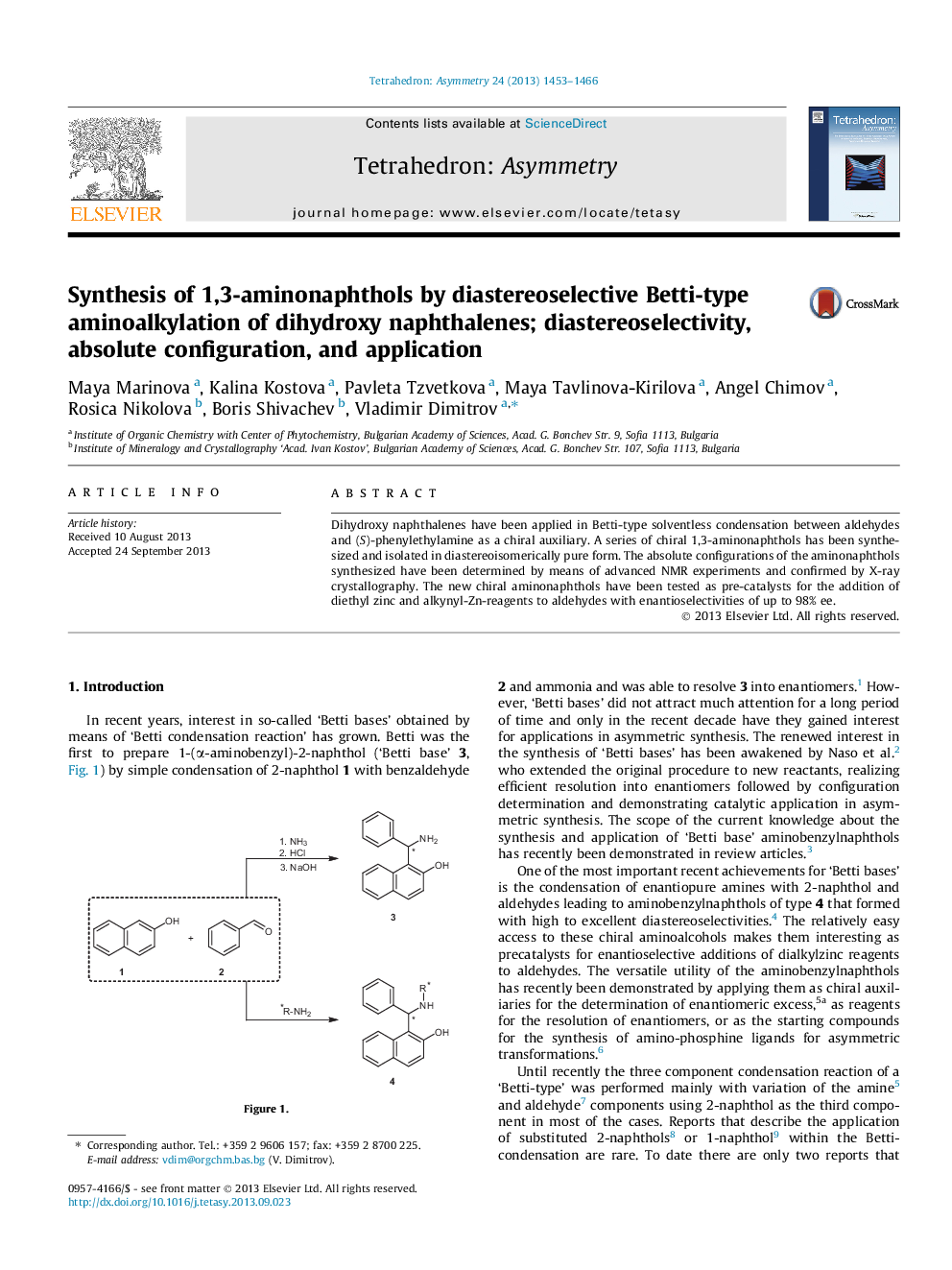
Dihydroxy naphthalenes have been applied in Betti-type solventless condensation between aldehydes and (S)-phenylethylamine as a chiral auxiliary. A series of chiral 1,3-aminonaphthols has been synthesized and isolated in diastereoisomerically pure form. The absolute configurations of the aminonaphthols synthesized have been determined by means of advanced NMR experiments and confirmed by X-ray crystallography. The new chiral aminonaphthols have been tested as pre-catalysts for the addition of diethyl zinc and alkynyl-Zn-reagents to aldehydes with enantioselectivities of up to 98% ee.
Figure optionsDownload as PowerPoint slide
1-((S)-((S)-1-Phenylethylamino)(m-tolyl)methyl)naphthalene-2,6-diolC26H25NO2[α]D20=+142.6 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
1-((S)-Naphthalen-1-yl((S)-1-phenylethylamino)methyl)naphthalene-2,6-diolC29H25NO2[α]D20=+522.9 (c 1.00, DMSO)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
1-((S)-((S)-1-Phenylethylamino)(m-tolyl)methyl)naphthalene-2,3-diolC26H25NO2[α]D20=+194.2 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
1-((S)-((S)-1-Phenylethylamino)(m-tolyl)methyl)naphthalen-2-olC26H25NO[α]D20=+197.0 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
1-((R)-((S)-1-Phenylethylamino)(m-tolyl)methyl)naphthalen-2-olC26H25NO[α]D20=-70.7 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (R,S)
1-((S)-Naphthalen-1-yl((S)-1-phenylethylamino)methyl)naphthalen-2-olC29H25NO[α]D20=+449.4 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
(S)-2-((S)-1-Phenylethyl)-1-m-tolyl-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazin-8-olC27H25NO2[α]D20=+129.4 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
(S)-1-(Naphthalen-1-yl)-2-((S)-1-phenylethyl)-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazin-8-olC30H25NO2[α]D20=+257.4 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
(S)-2-((S)-1-Phenylethyl)-1-m-tolyl-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazin-5-olC27H25NO2[α]D20=+131.4 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
(S)-2-((S)-1-Phenylethyl)-1-m-tolyl-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazineC27H25NO[α]D20=+164.7 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
(R)-2-((S)-1-Phenylethyl)-1-m-tolyl-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazineC27H25NO[α]D20=-64.8 (c 0.45, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (R,S)
(S)-1-(Naphthalen-1-yl)-2-((S)-1-phenylethyl)-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazineC30H25NO[α]D20=+283.7 (c 1.00, CHCl3)Source of chirality: (S)-phenylethylamineAbsolute configuration: (S,S)
Journal: Tetrahedron: Asymmetry - Volume 24, Issue 23, 15 December 2013, Pages 1453–1466