| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1346945 | 980286 | 2009 | 4 صفحه PDF | دانلود رایگان |
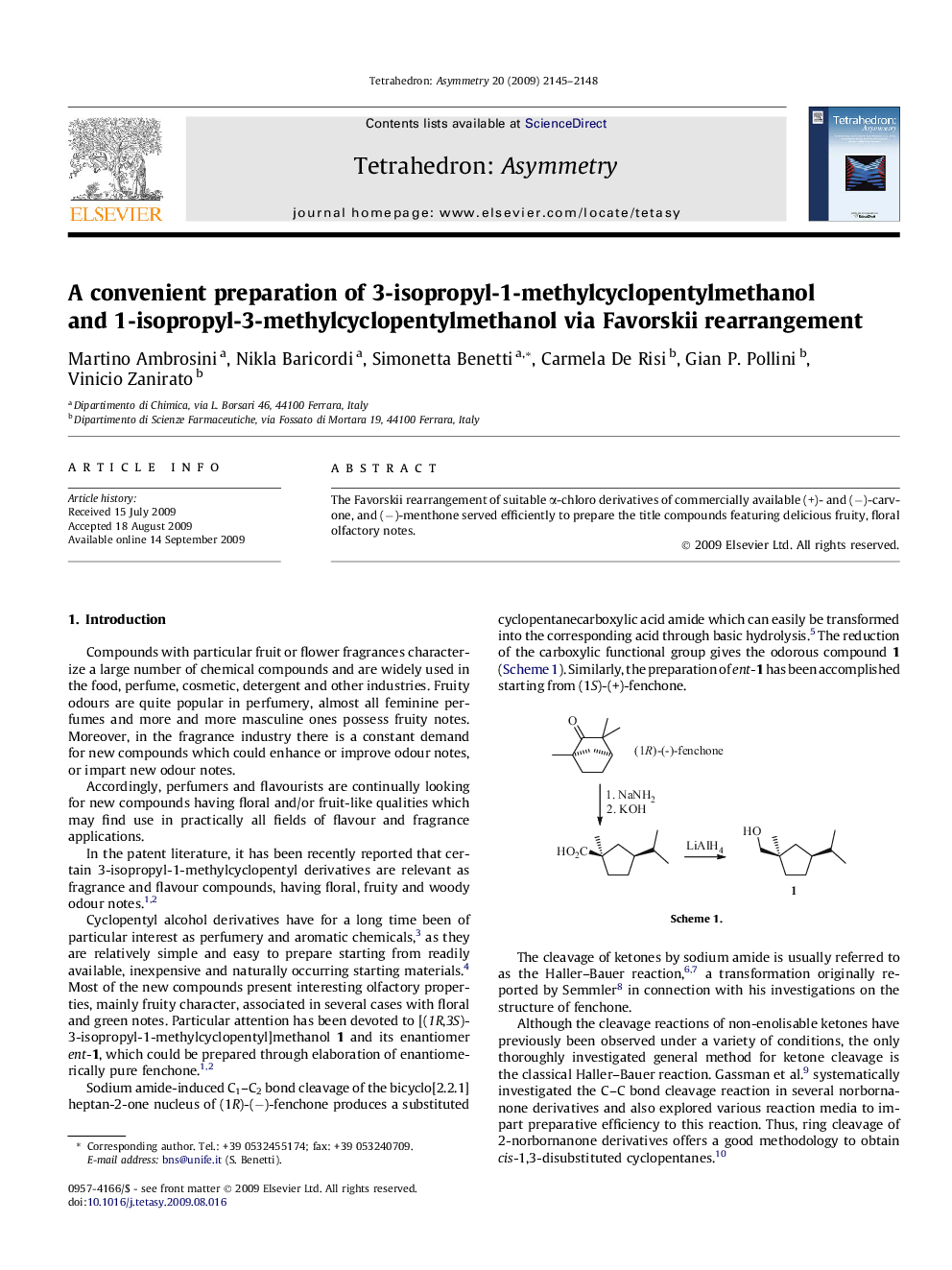
The Favorskii rearrangement of suitable α-chloro derivatives of commercially available (+)- and (−)-carvone, and (−)-menthone served efficiently to prepare the title compounds featuring delicious fruity, floral olfactory notes.
Figure optionsDownload as PowerPoint slide
[(1R,3R)-3-Methyl-1-isopropylcyclopentyl]-methanolC10H20O[α]D25=+10.9 (c 1, CHCl3)Source of chirality: (2S,5R)-(−)-menthoneAbsolute configuration: (1R,3R)
[(1R,3S)-3-Isopropyl-1-methylcyclopentyl]-methanolC10H20O[α]D25=-11.4 (c 1, CHCl3)Source of chirality: (S)-(+)-carvoneAbsolute configuration: (1R,3S)
Methyl (1R,3S)-3-isopropyl-1-methyl-cyclopentanecarboxylateC11H20O2[α]D25=-7.8 (c 1, CHCl3)Source of chirality: (S)-(+)-carvoneAbsolute configuration: (1R,3S)
Methyl (1S,3R)-1-isopropyl-3-methyl-cyclopentanecarboxylateC11H20O2[α]D25=-16.3 (c 1, CHCl3)Source of chirality: (2S,5R)-(−)-menthoneAbsolute configuration: (1S,3R)
Methyl (1R,3R)-1-isopropyl-3-methyl-cyclopentanecarboxylateC11H20O2[α]D25=+3.8 (c 1, CHCl3)Source of chirality: (2S,5R)-(−)-menthoneAbsolute configuration: (1R,3R)
[(1S,3R)-3-Methyl-1-isopropylcyclopentyl]-methanolC10H20O[α]D25=+15.6 (c 1, CHCl3)Source of chirality: (2S,5R)-(−)-menthoneAbsolute configuration: (1S,3R)
Journal: Tetrahedron: Asymmetry - Volume 20, Issue 18, 23 September 2009, Pages 2145–2148