| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1347254 | 980302 | 2009 | 6 صفحه PDF | دانلود رایگان |
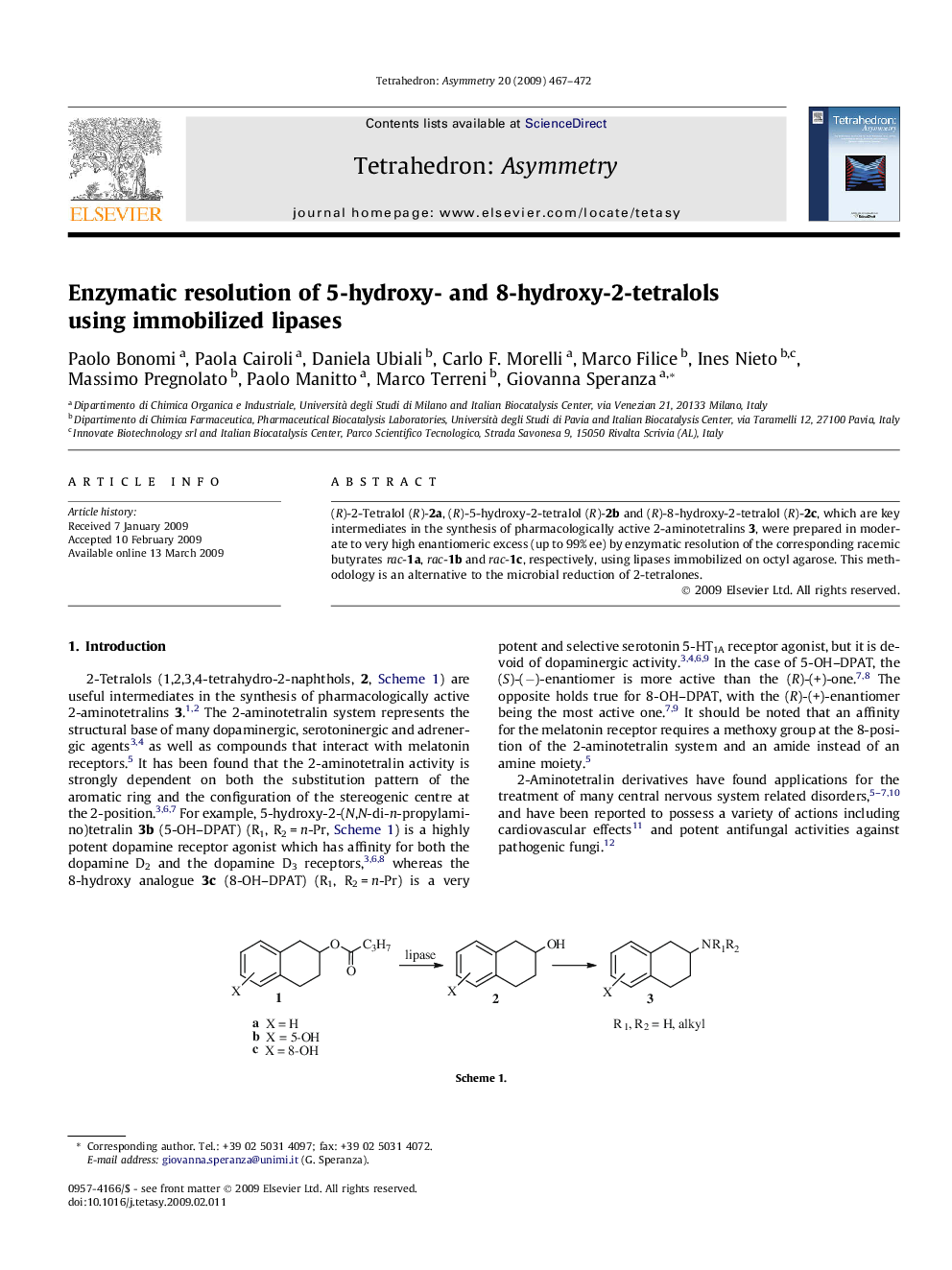
(R)-2-Tetralol (R)-2a, (R)-5-hydroxy-2-tetralol (R)-2b and (R)-8-hydroxy-2-tetralol (R)-2c, which are key intermediates in the synthesis of pharmacologically active 2-aminotetralins 3, were prepared in moderate to very high enantiomeric excess (up to 99% ee) by enzymatic resolution of the corresponding racemic butyrates rac-1a, rac-1b and rac-1c, respectively, using lipases immobilized on octyl agarose. This methodology is an alternative to the microbial reduction of 2-tetralones.
Figure optionsDownload as PowerPoint slide
(R)-8-Hydroxy-2-tetralolC10H12O2Ee = 99%[α]D25=+92.5 (c 0.5, MeOH)Source of chirality: enzymatic resolutionAbsolute configuration: (R)
(S)-8-Hydroxy-2-tetralyl butyrateC14H18O3Ee = 99%[α]D25=-39.2 (c 0.5, MeOH)Source of chirality: enzymatic resolutionAbsolute configuration: (S)
(R)-5-Hydroxy-2-tetralolC10H12O2Ee = 71%[α]D25=+50.6 (c 0.5, MeOH)Source of chirality: enzymatic resolutionAbsolute configuration: (R)
(S)-5-Hydroxy-2-tetralyl butyrateC14H18O3Ee = 65%[α]D25=-21.5 (c 0.5, MeOH)Source of chirality: enzymatic resolutionAbsolute configuration: (S)
Journal: Tetrahedron: Asymmetry - Volume 20, Issue 4, 11 March 2009, Pages 467–472