| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1347586 | 1500351 | 2013 | 5 صفحه PDF | دانلود رایگان |
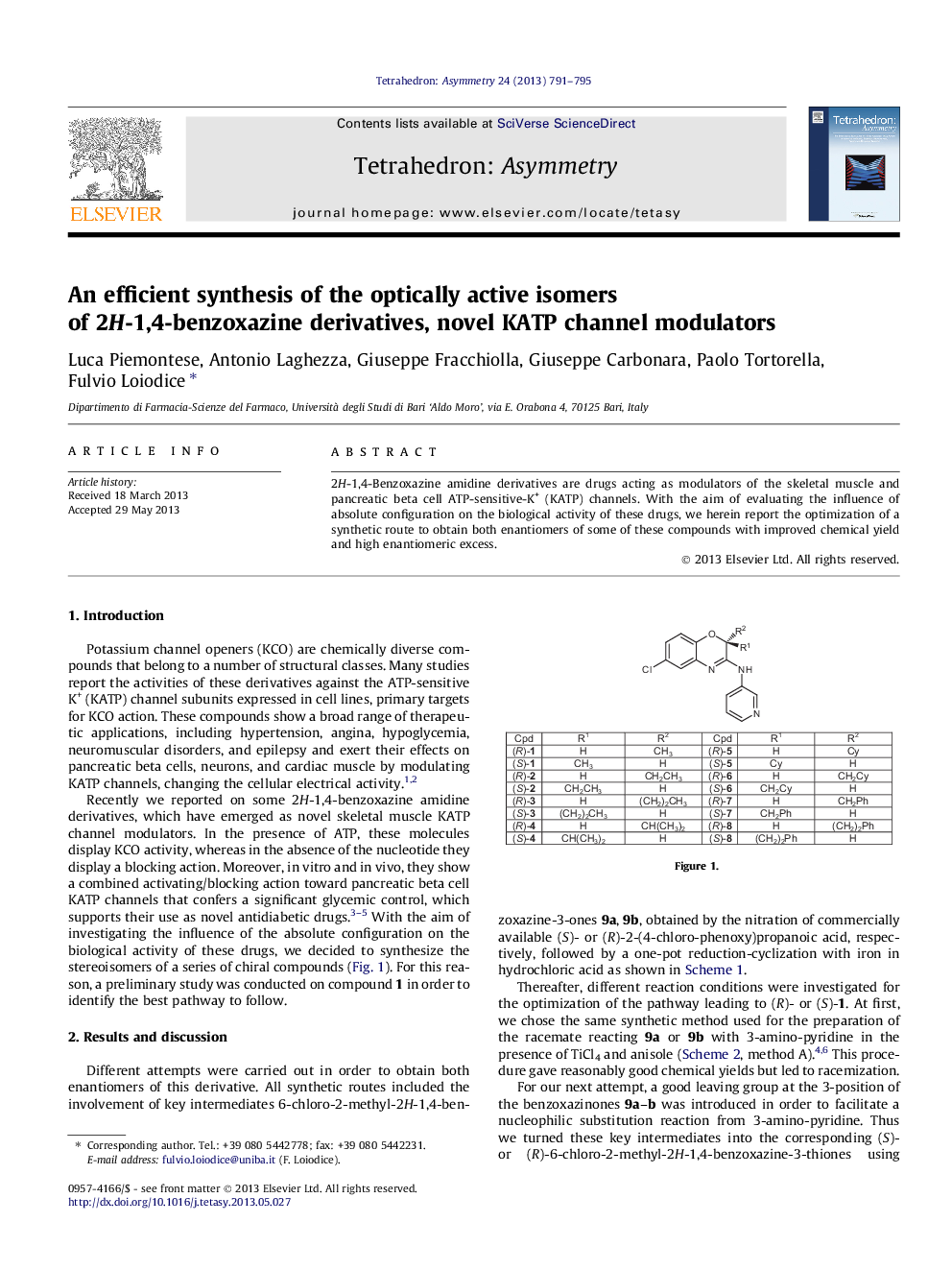
2H-1,4-Benzoxazine amidine derivatives are drugs acting as modulators of the skeletal muscle and pancreatic beta cell ATP-sensitive-K+ (KATP) channels. With the aim of evaluating the influence of absolute configuration on the biological activity of these drugs, we herein report the optimization of a synthetic route to obtain both enantiomers of some of these compounds with improved chemical yield and high enantiomeric excess.
Figure optionsDownload as PowerPoint slide
(S)-6-Chloro-2-methyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC14H12N3ClOEe = 97%[α]D20=-256 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-methyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-ethyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC15H14N3ClOEe = 94%[α]D20=-280 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-ethyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-propyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC16H16N3ClOEe = 97%[α]D20=-274 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-propyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-isopropyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC16H16N3ClOEe = 99%[α]D20=-373 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-isopropyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-cyclohexyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC19H20N3ClOEe = 99%[α]D20=-353 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-cyclohexyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-cyclohexylmethyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC20H22N3ClOEe = 99%[α]D20=-256 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-cyclohexylmethyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-2-Benzyl-6-chloro-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC20H16N3ClOEe = 99%[α]D20=-273 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-benzyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
(S)-6-Chloro-2-phenethyl-3-(pyridin-3-yl-amino)-2H-1,4-benzoxazineC21H18N3ClOEe = 99%[α]D20=-244 (c 1.0, MeOH)Source of chirality: (S)-6-Chloro-2-phenethyl-2H-1,4-benzoxazine-3-oneAbsolute configuration: (S)
Journal: Tetrahedron: Asymmetry - Volume 24, Issues 13–14, 31 July 2013, Pages 791–795