| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1349470 | 980400 | 2009 | 5 صفحه PDF | دانلود رایگان |
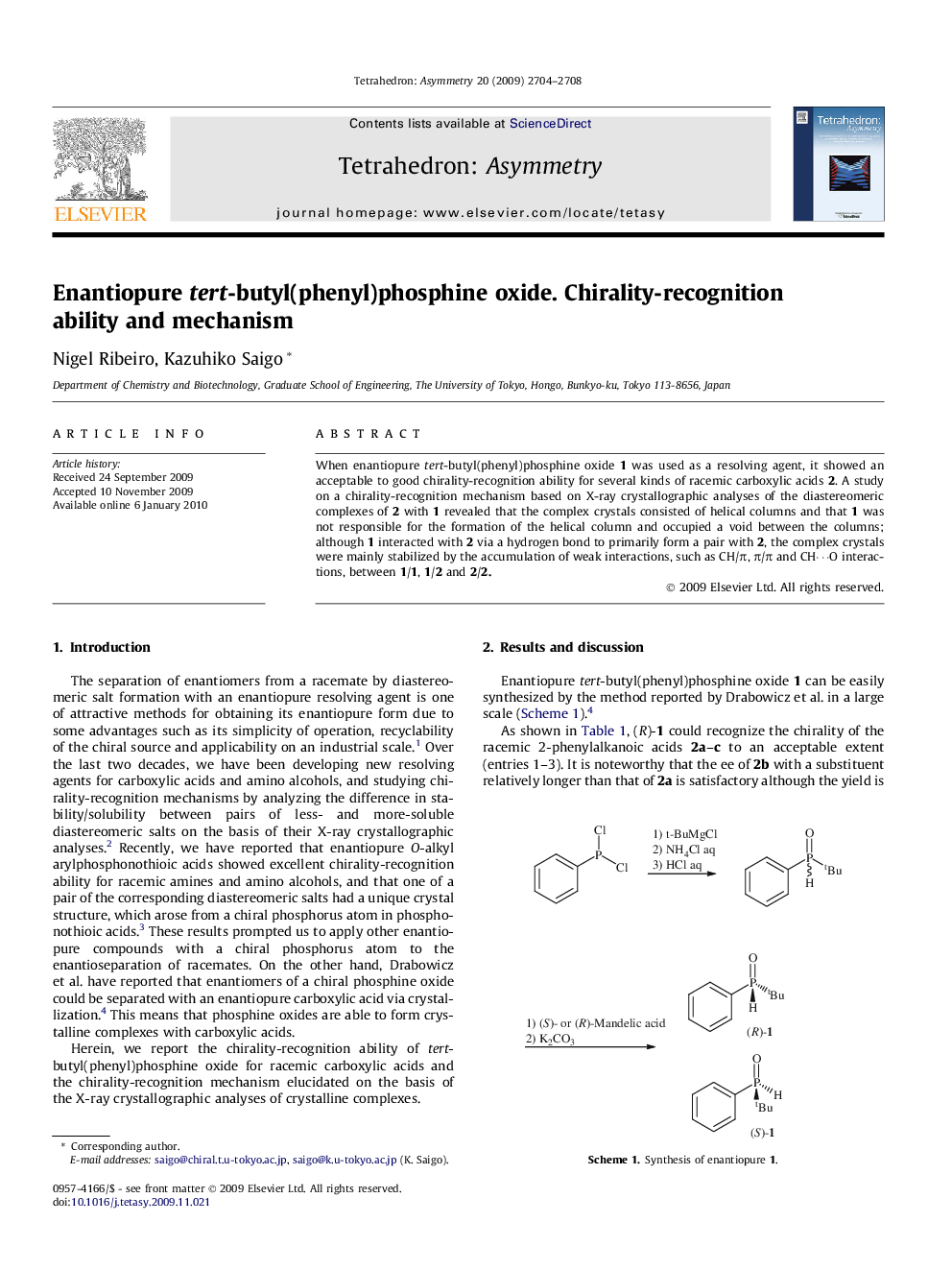
When enantiopure tert-butyl(phenyl)phosphine oxide 1 was used as a resolving agent, it showed an acceptable to good chirality-recognition ability for several kinds of racemic carboxylic acids 2. A study on a chirality-recognition mechanism based on X-ray crystallographic analyses of the diastereomeric complexes of 2 with 1 revealed that the complex crystals consisted of helical columns and that 1 was not responsible for the formation of the helical column and occupied a void between the columns; although 1 interacted with 2 via a hydrogen bond to primarily form a pair with 2, the complex crystals were mainly stabilized by the accumulation of weak interactions, such as CH/π, π/π and CH⋯O interactions, between 1/1, 1/2 and 2/2.
Figure optionsDownload as PowerPoint slide
Journal: Tetrahedron: Asymmetry - Volume 20, Issue 23, 11 December 2009, Pages 2704–2708