| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 179556 | 459355 | 2013 | 4 صفحه PDF | دانلود رایگان |
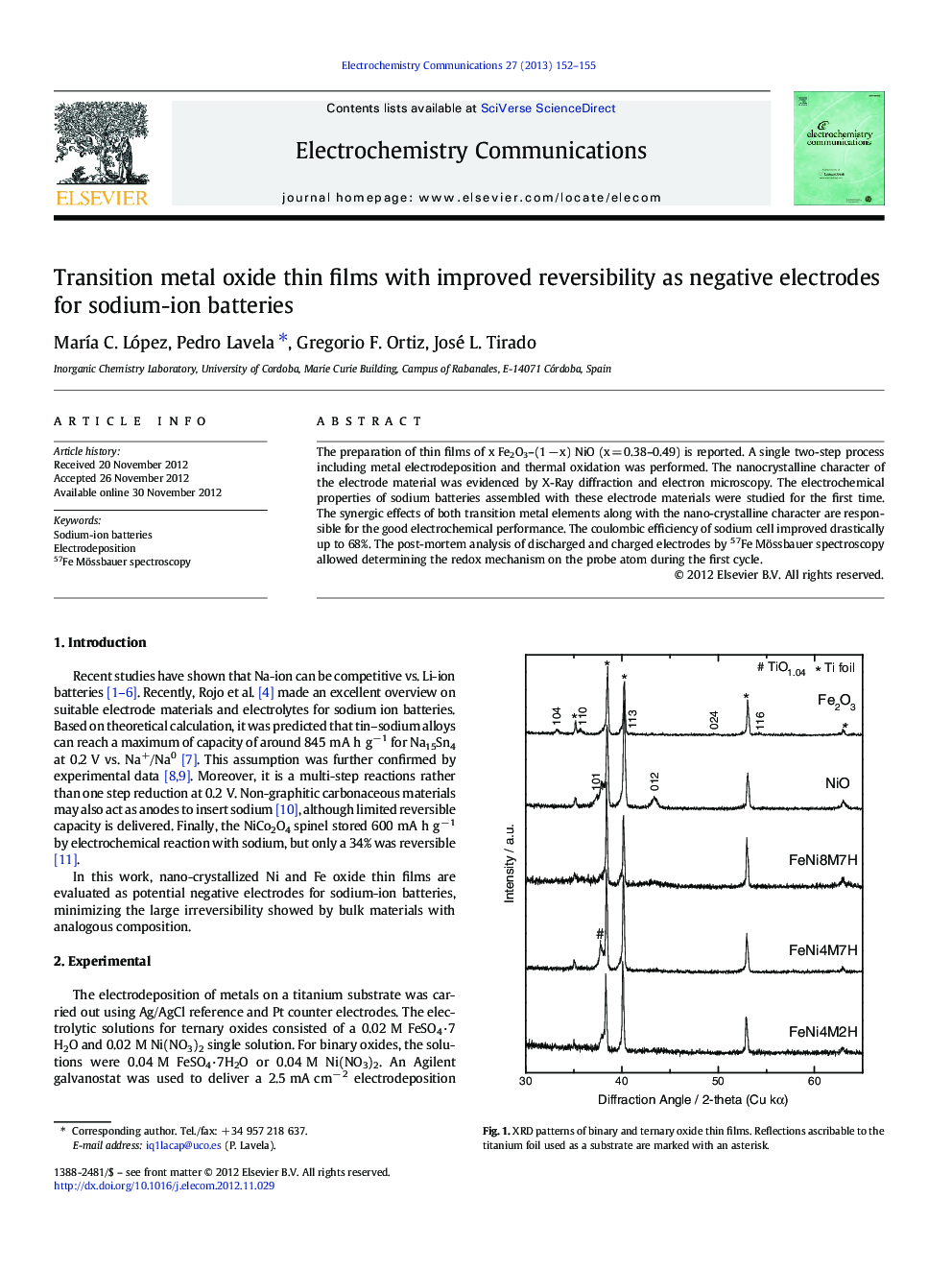
The preparation of thin films of x Fe2O3–(1 − x) NiO (x = 0.38–0.49) is reported. A single two-step process including metal electrodeposition and thermal oxidation was performed. The nanocrystalline character of the electrode material was evidenced by X-Ray diffraction and electron microscopy. The electrochemical properties of sodium batteries assembled with these electrode materials were studied for the first time. The synergic effects of both transition metal elements along with the nano-crystalline character are responsible for the good electrochemical performance. The coulombic efficiency of sodium cell improved drastically up to 68%. The post-mortem analysis of discharged and charged electrodes by 57Fe Mössbauer spectroscopy allowed determining the redox mechanism on the probe atom during the first cycle.
► Thin film conversion electrodes are prepared by a novel electrodeposition route.
► Polymer binders and conductive carbon additives are avoided.
► Coulombic inefficiency and cell polarization are lower than in bulk materials.
► Reversible capacity values are unusually high for sodium cells.
Journal: Electrochemistry Communications - Volume 27, February 2013, Pages 152–155