| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 59015 | 1419445 | 2014 | 7 صفحه PDF | دانلود رایگان |
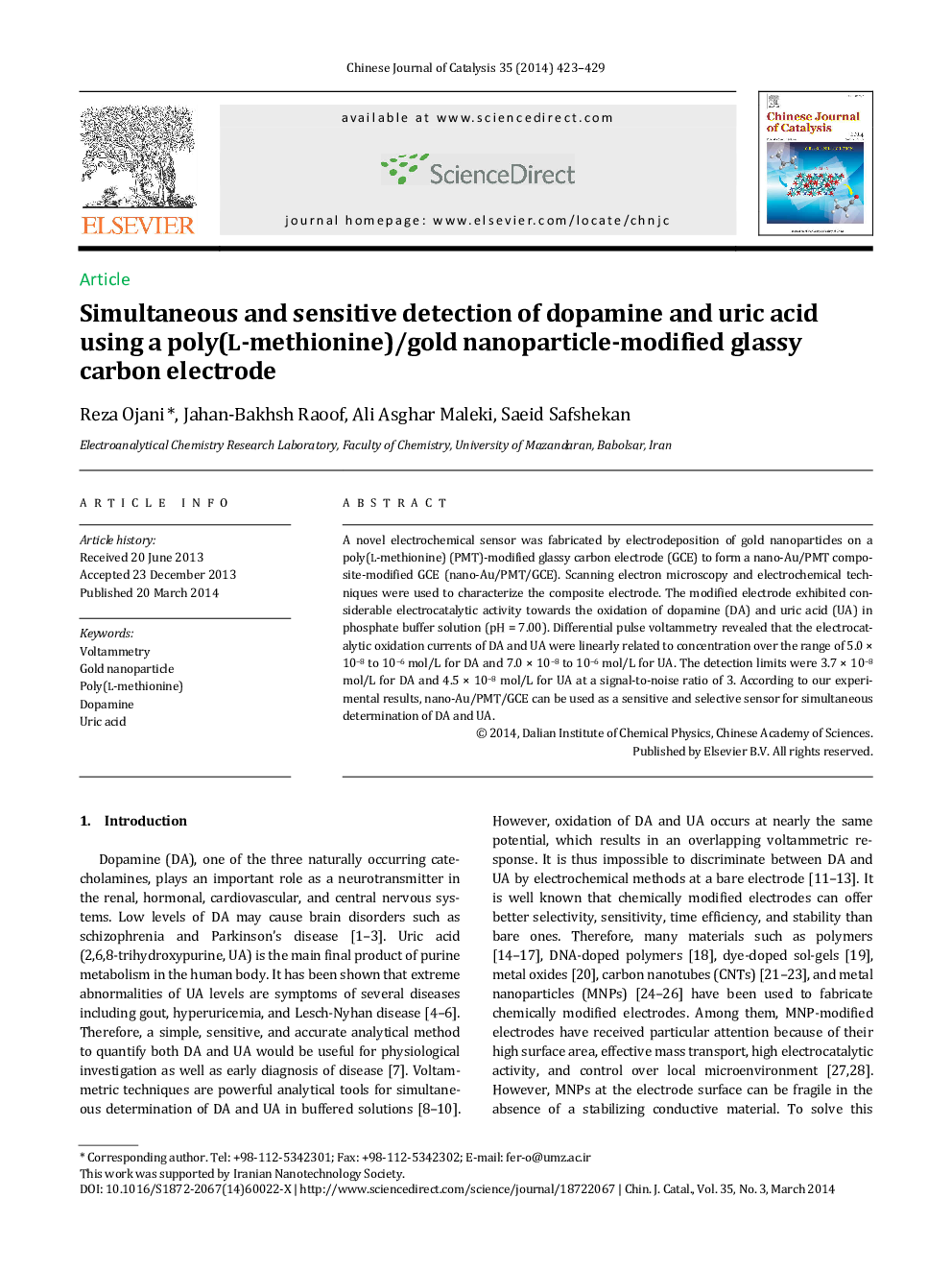
A novel electrochemical sensor was fabricated by electrodeposition of gold nanoparticles on a poly(L-methionine) (PMT)-modified glassy carbon electrode (GCE) to form a nano-Au/PMT composite-modified GCE (nano-Au/PMT/GCE). Scanning electron microscopy and electrochemical techniques were used to characterize the composite electrode. The modified electrode exhibited considerable electrocatalytic activity towards the oxidation of dopamine (DA) and uric acid (UA) in phosphate buffer solution (pH = 7.00). Differential pulse voltammetry revealed that the electrocatalytic oxidation currents of DA and UA were linearly related to concentration over the range of 5.0 × 10−8 to 10−6 mol/L for DA and 7.0 × 10−8 to 10−6 mol/L for UA. The detection limits were 3.7 × 10−8 mol/L for DA and 4.5 × 10−8 mol/L for UA at a signal-to-noise ratio of 3. According to our experimental results, nano-Au/PMT/GCE can be used as a sensitive and selective sensor for simultaneous determination of DA and UA.
Graphical AbstractA glassy carbon electrode modified with gold nanoparticles and poly(L-methionine) exhibited high electrocatalytic activity towards the oxidation of dopamine and uric acid. This electrode allowed sensitive and selective determination of dopamine and uric acid at nanomolar levels.Figure optionsDownload as PowerPoint slide
Journal: Chinese Journal of Catalysis - Volume 35, Issue 3, March 2014, Pages 423–429