| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229036 | 1495231 | 2015 | 9 صفحه PDF | دانلود رایگان |
• Two Schiff bases were successfully synthesized by a facile approach.
• These were characterized by 1H NMR, 13C NMR, FTIR and UV–Vis spectroscopy.
• The pH dependent redox and photometric behavior was investigated in a wide pH range.
• The experimental findings were supported by quantum mechanical approach.
• Isosbestic points indicated the existence of Schiff bases in different tautomeric forms.
A new Schiff base, 1-((4-bromophenylimino) methyl) naphthalen-2-ol (BPIMN) was successfully synthesized and characterized by 1H NMR, 13C NMR, FTIR and UV–Vis spectroscopy. The results were compared with a structurally related Schiff base, 1-((4-chlorophenylimino) methyl) naphthalen-2-ol (CPIMN). The photometric and electrochemical fate of BPIMN and CPIMN was investigated in a wide pH range. The experimental findings were supported by quantum mechanical approach. The redox mechanistic pathways were proposed on the basis of results obtained electrochemical techniques. Moreover, pH dependent UV–Vis spectroscopy of BPIMN and CPIMN was carried out and the appearance of isosbestic points indicated the existence of these compounds in different tautomeric forms.
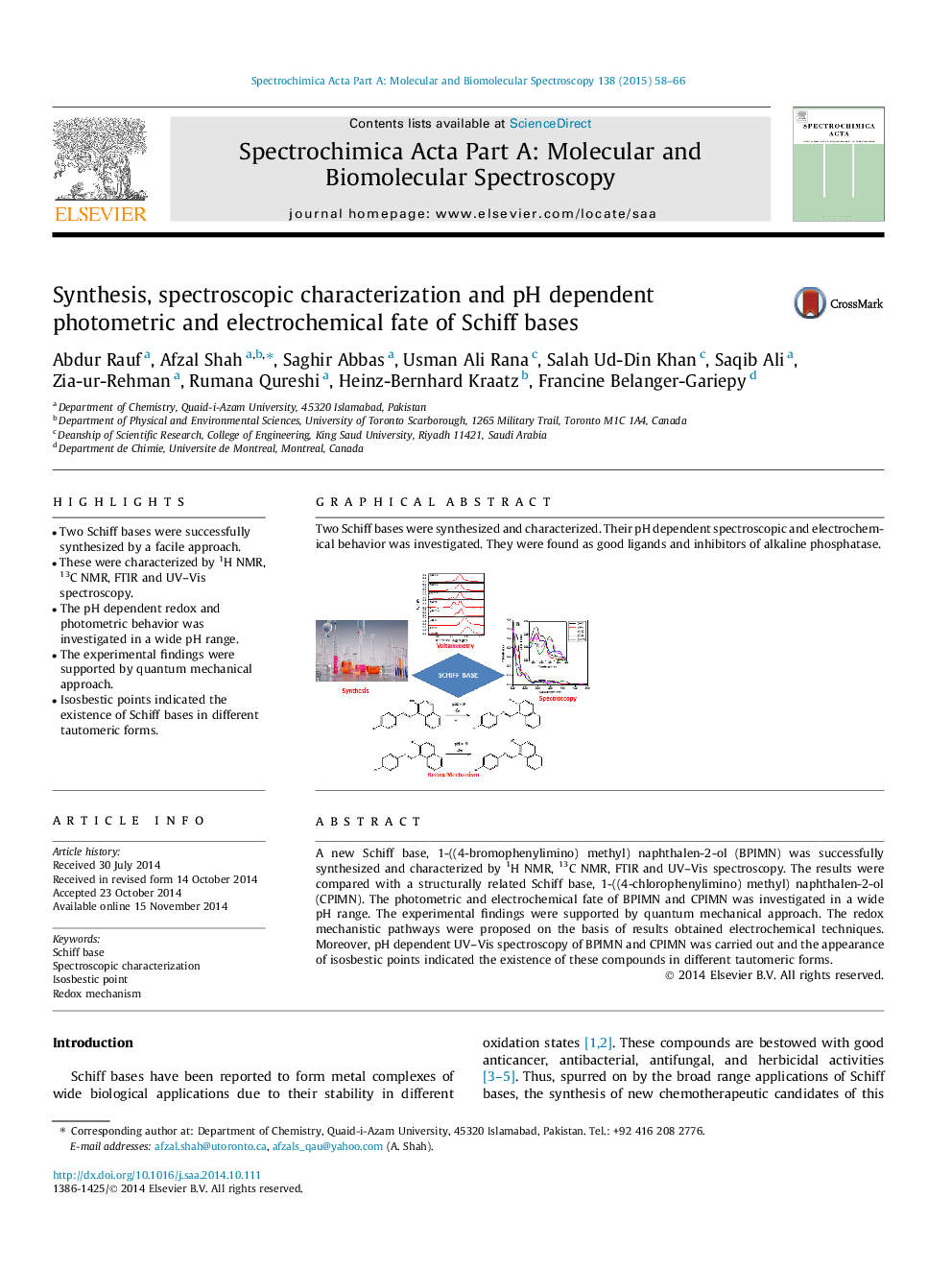
Two Schiff bases were synthesized and characterized. Their pH dependent spectroscopic and electrochemical behavior was investigated. They were found as good ligands and inhibitors of alkaline phosphatase.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 138, 5 March 2015, Pages 58–66