| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229093 | 1495231 | 2015 | 10 صفحه PDF | دانلود رایگان |
• Novel metal complexes of (N4) macrocyclic ligand have been synthesized and characterized.
• Analytical, spectral and thermal data confirm the structure of the compounds.
• In vitro antitumor activity shows that the obtained compounds are potent anticancer agents.
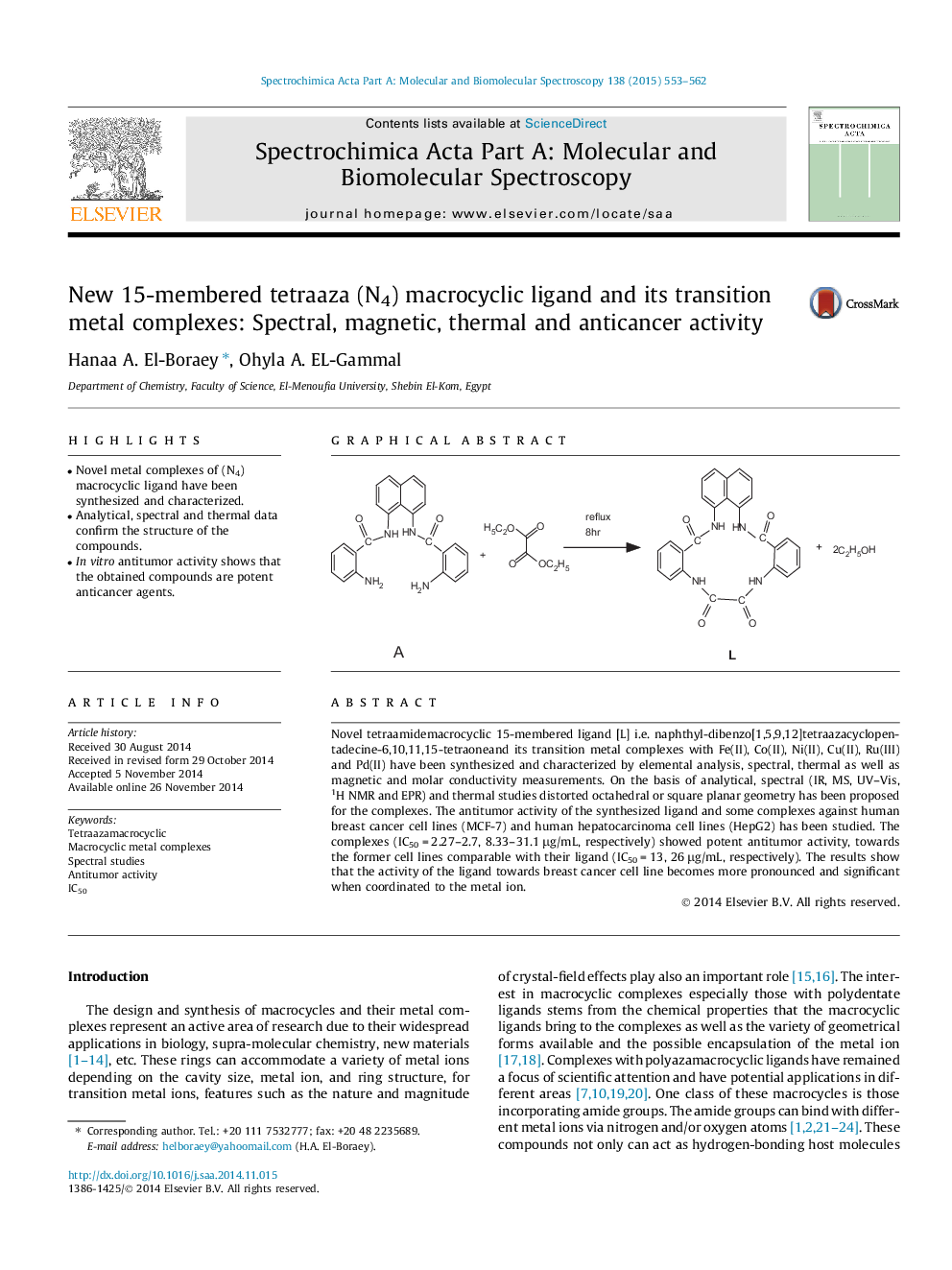
Novel tetraamidemacrocyclic 15-membered ligand [L] i.e. naphthyl-dibenzo[1,5,9,12]tetraazacyclopentadecine-6,10,11,15-tetraoneand its transition metal complexes with Fe(II), Co(II), Ni(II), Cu(II), Ru(III) and Pd(II) have been synthesized and characterized by elemental analysis, spectral, thermal as well as magnetic and molar conductivity measurements. On the basis of analytical, spectral (IR, MS, UV–Vis, 1H NMR and EPR) and thermal studies distorted octahedral or square planar geometry has been proposed for the complexes. The antitumor activity of the synthesized ligand and some complexes against human breast cancer cell lines (MCF-7) and human hepatocarcinoma cell lines (HepG2) has been studied. The complexes (IC50 = 2.27–2.7, 8.33–31.1 μg/mL, respectively) showed potent antitumor activity, towards the former cell lines comparable with their ligand (IC50 = 13, 26 μg/mL, respectively). The results show that the activity of the ligand towards breast cancer cell line becomes more pronounced and significant when coordinated to the metal ion.
Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 138, 5 March 2015, Pages 553–562