| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229502 | 968734 | 2015 | 7 صفحه PDF | دانلود رایگان |
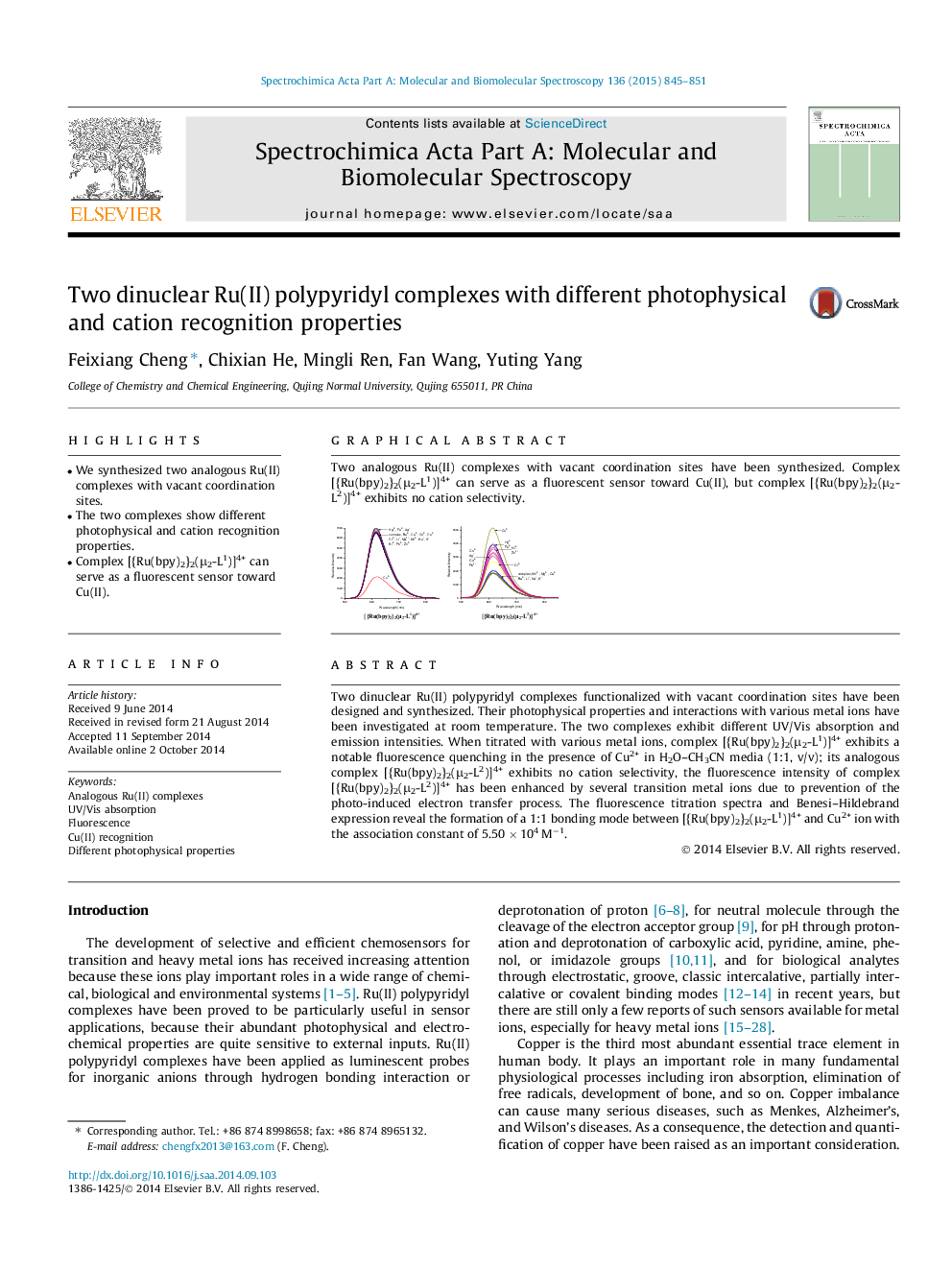
• We synthesized two analogous Ru(II) complexes with vacant coordination sites.
• The two complexes show different photophysical and cation recognition properties.
• Complex [{Ru(bpy)2}2(μ2-L1)]4+ can serve as a fluorescent sensor toward Cu(II).
Two dinuclear Ru(II) polypyridyl complexes functionalized with vacant coordination sites have been designed and synthesized. Their photophysical properties and interactions with various metal ions have been investigated at room temperature. The two complexes exhibit different UV/Vis absorption and emission intensities. When titrated with various metal ions, complex [{Ru(bpy)2}2(μ2-L1)]4+ exhibits a notable fluorescence quenching in the presence of Cu2+ in H2O–CH3CN media (1:1, v/v); its analogous complex [{Ru(bpy)2}2(μ2-L2)]4+ exhibits no cation selectivity, the fluorescence intensity of complex [{Ru(bpy)2}2(μ2-L2)]4+ has been enhanced by several transition metal ions due to prevention of the photo-induced electron transfer process. The fluorescence titration spectra and Benesi–Hildebrand expression reveal the formation of a 1:1 bonding mode between [{Ru(bpy)2}2(μ2-L1)]4+ and Cu2+ ion with the association constant of 5.50 × 104 M−1.
Two analogous Ru(II) complexes with vacant coordination sites have been synthesized. Complex [{Ru(bpy)2}2(μ2-L1)]4+ can serve as a fluorescent sensor toward Cu(II), but complex [{Ru(bpy)2}2(μ2-L2)]4+ exhibits no cation selectivity.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 136, Part B, 5 February 2015, Pages 845–851