| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229692 | 1495234 | 2015 | 5 صفحه PDF | دانلود رایگان |
• A red phosphor CaAl4O7:Eu3+ has been synthesized by Pechini method.
• The photoluminescence properties were found to be influenced by the calcination temperature of the phosphors.
• The excitation wavelength and the narrow emission properties suggest their potential application in near-UV white LEDS.
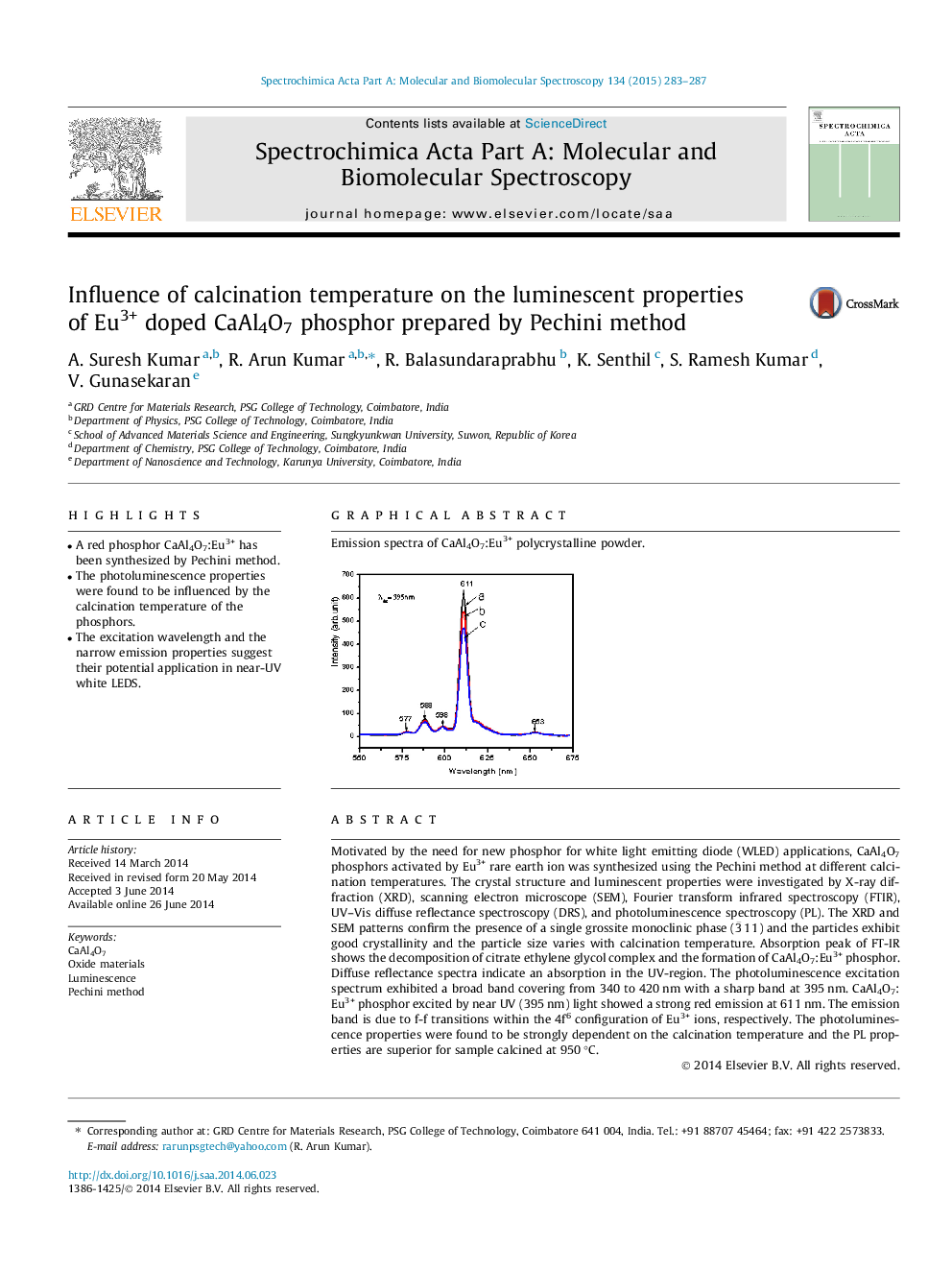
Motivated by the need for new phosphor for white light emitting diode (WLED) applications, CaAl4O7 phosphors activated by Eu3+ rare earth ion was synthesized using the Pechini method at different calcination temperatures. The crystal structure and luminescent properties were investigated by X-ray diffraction (XRD), scanning electron microscope (SEM), Fourier transform infrared spectroscopy (FTIR), UV–Vis diffuse reflectance spectroscopy (DRS), and photoluminescence spectroscopy (PL). The XRD and SEM patterns confirm the presence of a single grossite monoclinic phase (3¯11) and the particles exhibit good crystallinity and the particle size varies with calcination temperature. Absorption peak of FT-IR shows the decomposition of citrate ethylene glycol complex and the formation of CaAl4O7:Eu3+ phosphor. Diffuse reflectance spectra indicate an absorption in the UV-region. The photoluminescence excitation spectrum exhibited a broad band covering from 340 to 420 nm with a sharp band at 395 nm. CaAl4O7:Eu3+ phosphor excited by near UV (395 nm) light showed a strong red emission at 611 nm. The emission band is due to f-f transitions within the 4f6 configuration of Eu3+ ions, respectively. The photoluminescence properties were found to be strongly dependent on the calcination temperature and the PL properties are superior for sample calcined at 950 °C.
Emission spectra of CaAl4O7:Eu3+ polycrystalline powder.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 134, 5 January 2015, Pages 283–287