| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1230128 | 1495218 | 2015 | 6 صفحه PDF | دانلود رایگان |
• Sodium hexadecyl sulfate (SHS) was successed to be intercalated into the hydrocalumites, which was investigated by XRD, SEM combining with MIR and NIR spectroscopic methods.
• SHS could modify hydrocalumites to form organo-LDH.
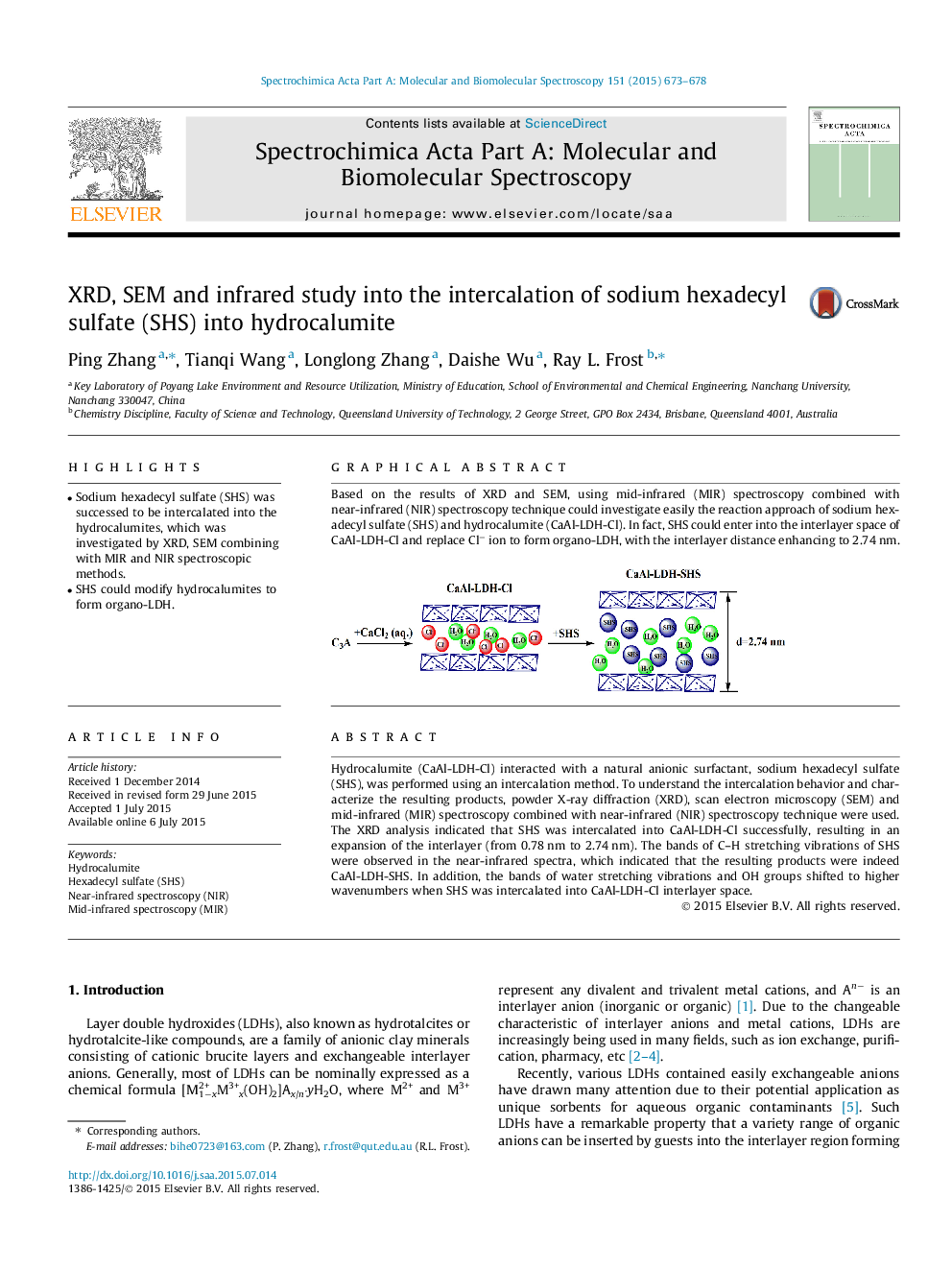
Hydrocalumite (CaAl-LDH-Cl) interacted with a natural anionic surfactant, sodium hexadecyl sulfate (SHS), was performed using an intercalation method. To understand the intercalation behavior and characterize the resulting products, powder X-ray diffraction (XRD), scan electron microscopy (SEM) and mid-infrared (MIR) spectroscopy combined with near-infrared (NIR) spectroscopy technique were used. The XRD analysis indicated that SHS was intercalated into CaAl-LDH-Cl successfully, resulting in an expansion of the interlayer (from 0.78 nm to 2.74 nm). The bands of C–H stretching vibrations of SHS were observed in the near-infrared spectra, which indicated that the resulting products were indeed CaAl-LDH-SHS. In addition, the bands of water stretching vibrations and OH groups shifted to higher wavenumbers when SHS was intercalated into CaAl-LDH-Cl interlayer space.
Based on the results of XRD and SEM, using mid-infrared (MIR) spectroscopy combined with near-infrared (NIR) spectroscopy technique could investigate easily the reaction approach of sodium hexadecyl sulfate (SHS) and hydrocalumite (CaAl-LDH-Cl). In fact, SHS could enter into the interlayer space of CaAl-LDH-Cl and replace Cl− ion to form organo-LDH, with the interlayer distance enhancing to 2.74 nm.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 151, 5 December 2015, Pages 673–678