| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1230869 | 1495248 | 2014 | 9 صفحه PDF | دانلود رایگان |
• Synthesis of a new quatridentate bis-amide ligand and its complexes.
• Characteristic NMR for the E/Z isomers of secondary amide ligand.
• Synthesis of good DNA intercalators.
• Appreciable redox behavior for Cu(II) complex.
A quatridentate bis-amide ligand, N,N′-propylenebis(salicylamide) H2pbs, and its transition metal complexes [M(pbs)(H2O)2], where M = Co(II), Ni(II), Cu(II), and Zn(II) have been synthesized and characterized by elemental analysis, UV–Vis, IR, NMR, Mass, EPR, molar conductivity, magnetic moment values and thermal analysis. The NMR spectrum of ligand evidences the E/Z isomerism. All the evidences reveal that the metal ions adopt octahedral geometry with metal:ligand:solvent ratio 1:1:2. The conductivity measurements exhibit that the complexes are non-electrolytes. DNA binding properties of these complexes have been explored by UV–Vis and cyclic voltammetry. The results indicate that these complexes are good intercalators.
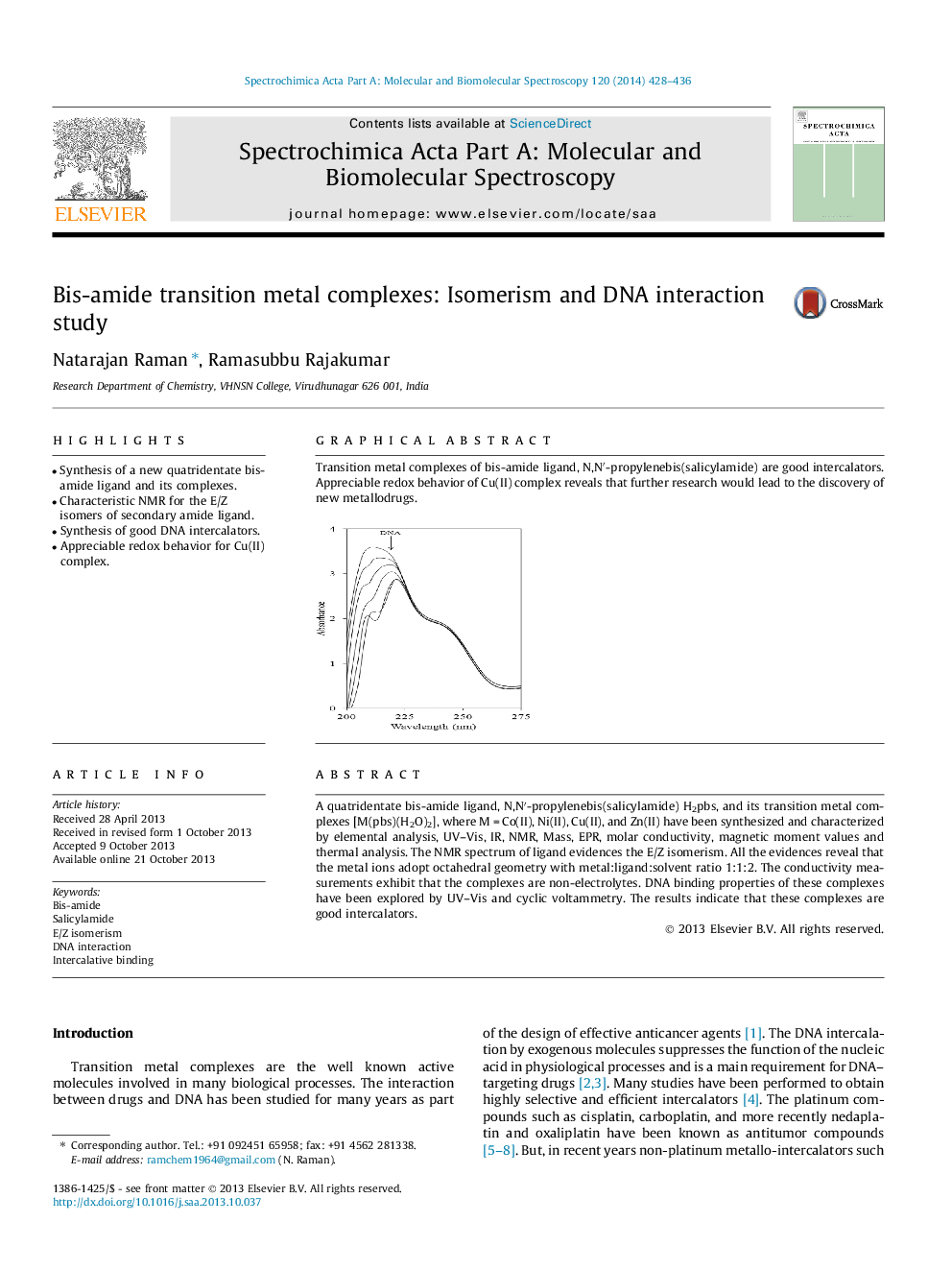
Transition metal complexes of bis-amide ligand, N,N′-propylenebis(salicylamide) are good intercalators. Appreciable redox behavior of Cu(II) complex reveals that further research would lead to the discovery of new metallodrugs.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 120, 24 February 2014, Pages 428–436