| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1232023 | 1495265 | 2013 | 7 صفحه PDF | دانلود رایگان |
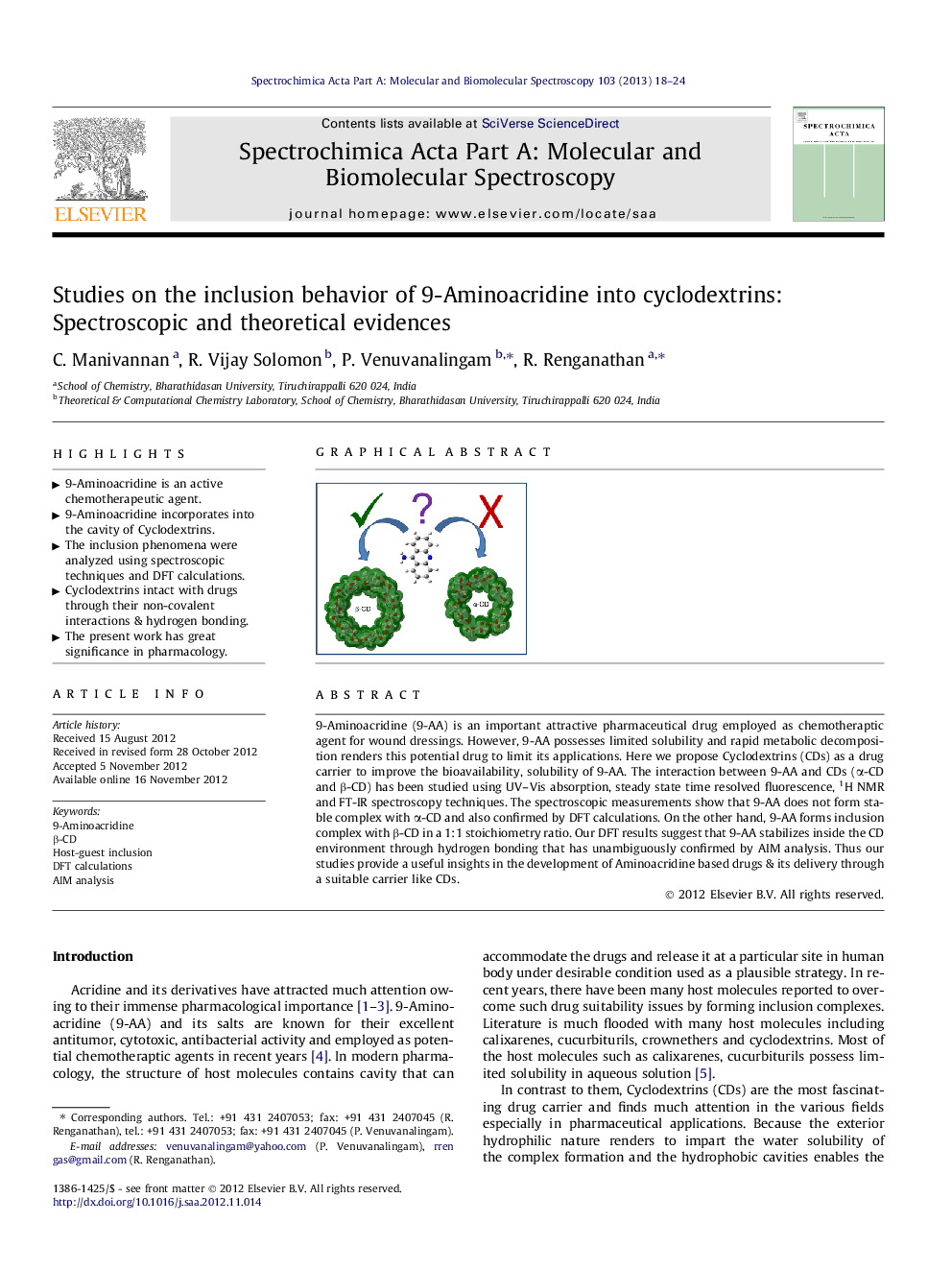
9-Aminoacridine (9-AA) is an important attractive pharmaceutical drug employed as chemotheraptic agent for wound dressings. However, 9-AA possesses limited solubility and rapid metabolic decomposition renders this potential drug to limit its applications. Here we propose cyclodextrins (CDs) as a drug carrier to improve the bioavailability, solubility of 9-AA. The interaction between 9-AA and CDs (α-CD and β-CD) has been studied using UV–Vis absorption, steady state time resolved fluorescence, 1H NMR and FT-IR spectroscopy techniques. The spectroscopic measurements show that 9-AA does not form stable complex with α-CD and also confirmed by DFT calculations. On the other hand, 9-AA forms inclusion complex with β-CD in a 1:1 stoichiometry ratio. Our DFT results suggest that 9-AA stabilizes inside the CD environment through hydrogen bonding that has unambiguously confirmed by AIM analysis. Thus our studies provide a useful insights in the development of Aminoacridine based drugs & its delivery through a suitable carrier like CDs.
Figure optionsDownload as PowerPoint slideHighlights
► 9-Aminoacridine is an active chemotherapeutic agent.
► 9-Aminoacridine incorporates into the cavity of cyclodextrins.
► The inclusion phenomena were analyzed using spectroscopic techniques and DFT calculations.
► Cyclodextrins intact with drugs through their non-covalent interactions and hydrogen bonding.
► The present work has great significance in pharmacology.
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 103, 15 February 2013, Pages 18–24