| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1233488 | 1495235 | 2014 | 12 صفحه PDF | دانلود رایگان |
• Synthesized Cu(II) Schiff base complexes were characterized with spectroscopy techniques.
• Thermodynamic parameters were measured spectrophotometrically.
• The DFT calculations were carried out to determine the geometrical properties.
• The molecular has been investigated by computational studies.
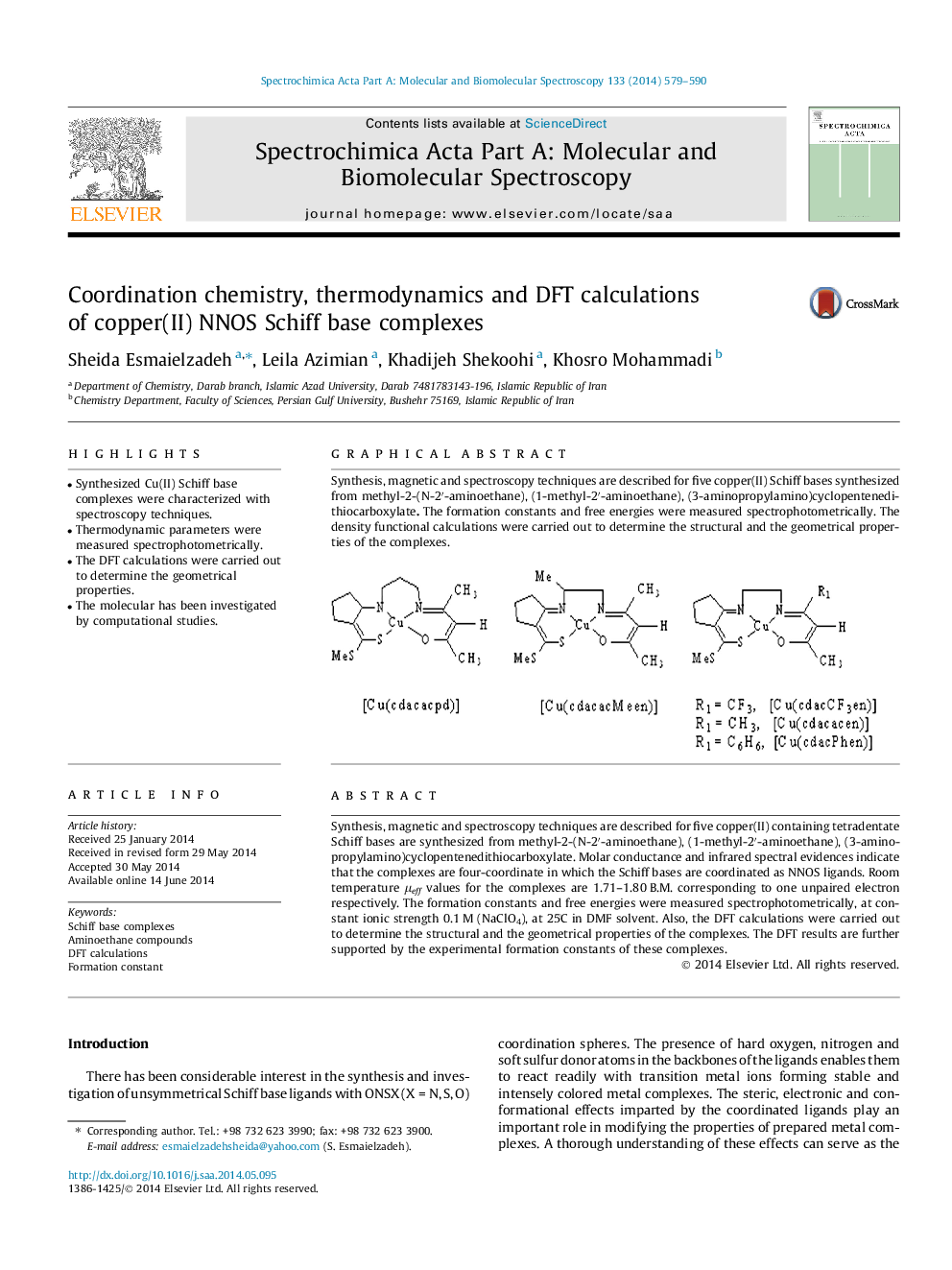
Synthesis, magnetic and spectroscopy techniques are described for five copper(II) containing tetradentate Schiff bases are synthesized from methyl-2-(N-2′-aminoethane), (1-methyl-2′-aminoethane), (3-aminopropylamino)cyclopentenedithiocarboxylate. Molar conductance and infrared spectral evidences indicate that the complexes are four-coordinate in which the Schiff bases are coordinated as NNOS ligands. Room temperature μeff values for the complexes are 1.71–1.80 B.M. corresponding to one unpaired electron respectively. The formation constants and free energies were measured spectrophotometrically, at constant ionic strength 0.1 M (NaClO4), at 25˚C in DMF solvent. Also, the DFT calculations were carried out to determine the structural and the geometrical properties of the complexes. The DFT results are further supported by the experimental formation constants of these complexes.
Synthesis, magnetic and spectroscopy techniques are described for five copper(II) Schiff bases synthesized from methyl-2-(N-2′-aminoethane), (1-methyl-2′-aminoethane), (3-aminopropylamino)cyclopentenedithiocarboxylate. The formation constants and free energies were measured spectrophotometrically. The density functional calculations were carried out to determine the structural and the geometrical properties of the complexes.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 133, 10 December 2014, Pages 579–590