| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1233977 | 1495241 | 2014 | 6 صفحه PDF | دانلود رایگان |
• Mixed-ligands [Cu(dipn)(2N)]Br2 complexes are synthesized.
• The complexes have significant antimicrobial and antiproliferative activity.
• The electronic spectra of the complexes are explained by DFT and TDDFT.
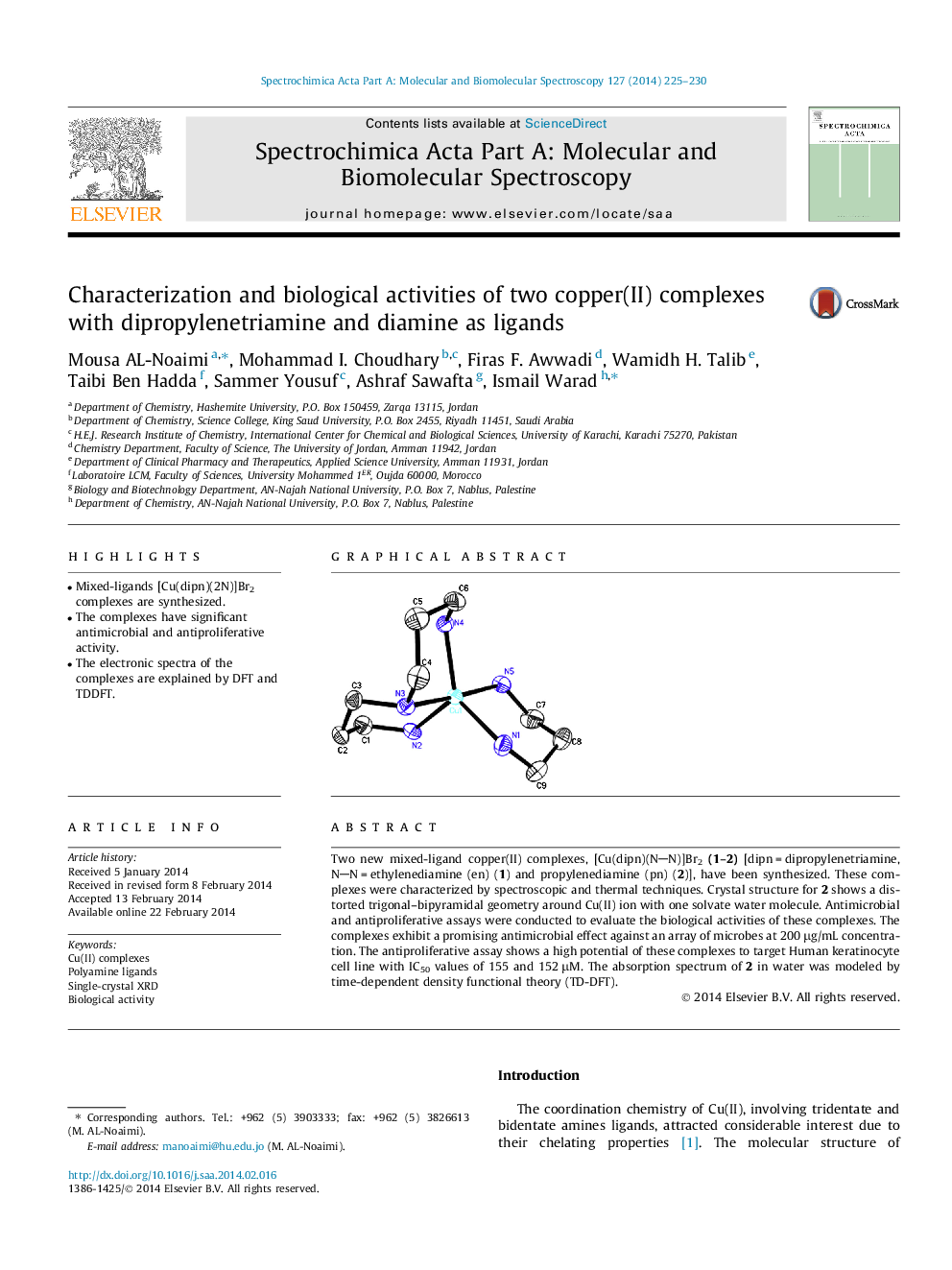
Two new mixed-ligand copper(II) complexes, [Cu(dipn)(NN)]Br2(1–2) [dipn = dipropylenetriamine, NN = ethylenediamine (en) (1) and propylenediamine (pn) (2)], have been synthesized. These complexes were characterized by spectroscopic and thermal techniques. Crystal structure for 2 shows a distorted trigonal–bipyramidal geometry around Cu(II) ion with one solvate water molecule. Antimicrobial and antiproliferative assays were conducted to evaluate the biological activities of these complexes. The complexes exhibit a promising antimicrobial effect against an array of microbes at 200 μg/mL concentration. The antiproliferative assay shows a high potential of these complexes to target Human keratinocyte cell line with IC50 values of 155 and 152 μM. The absorption spectrum of 2 in water was modeled by time-dependent density functional theory (TD-DFT).
Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 127, 5 June 2014, Pages 225–230