| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1235049 | 1495267 | 2013 | 6 صفحه PDF | دانلود رایگان |
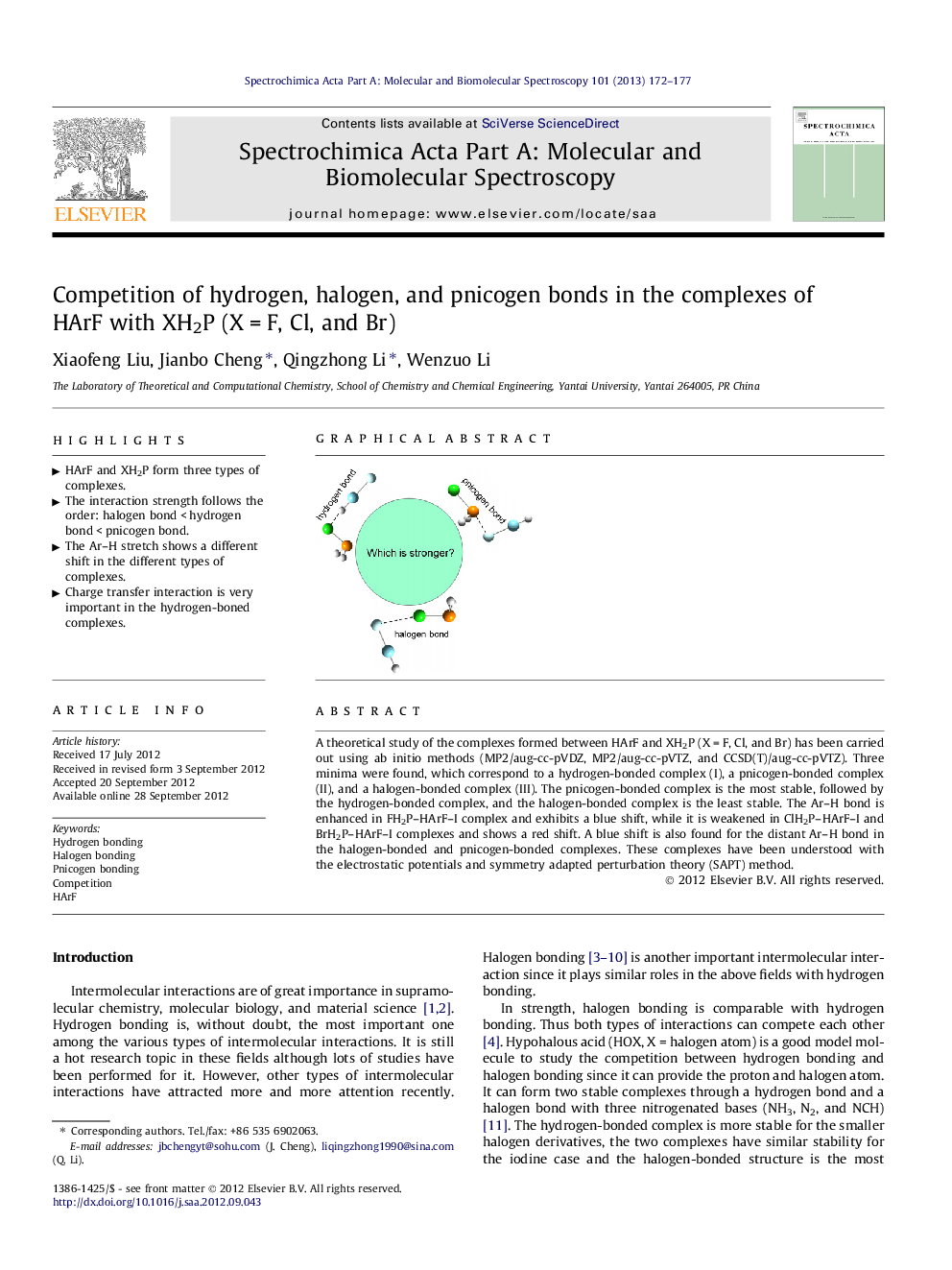
A theoretical study of the complexes formed between HArF and XH2P (X = F, Cl, and Br) has been carried out using ab initio methods (MP2/aug-cc-pVDZ, MP2/aug-cc-pVTZ, and CCSD(T)/aug-cc-pVTZ). Three minima were found, which correspond to a hydrogen-bonded complex (I), a pnicogen-bonded complex (II), and a halogen-bonded complex (III). The pnicogen-bonded complex is the most stable, followed by the hydrogen-bonded complex, and the halogen-bonded complex is the least stable. The Ar–H bond is enhanced in FH2P–HArF–I complex and exhibits a blue shift, while it is weakened in ClH2P–HArF–I and BrH2P–HArF–I complexes and shows a red shift. A blue shift is also found for the distant Ar–H bond in the halogen-bonded and pnicogen-bonded complexes. These complexes have been understood with the electrostatic potentials and symmetry adapted perturbation theory (SAPT) method.
Figure optionsDownload as PowerPoint slideHighlights
► HArF and XH2P form three types of complexes.
► The interaction strength follows the order: halogen bond < hydrogen bond < pnicogen bond.
► The Ar–H stretch shows a different shift in the different types of complexes.
► Charge transfer interaction is very important in the hydrogen-boned complexes.
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 101, 15 January 2013, Pages 172–177