| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1369133 | 981764 | 2013 | 4 صفحه PDF | دانلود رایگان |
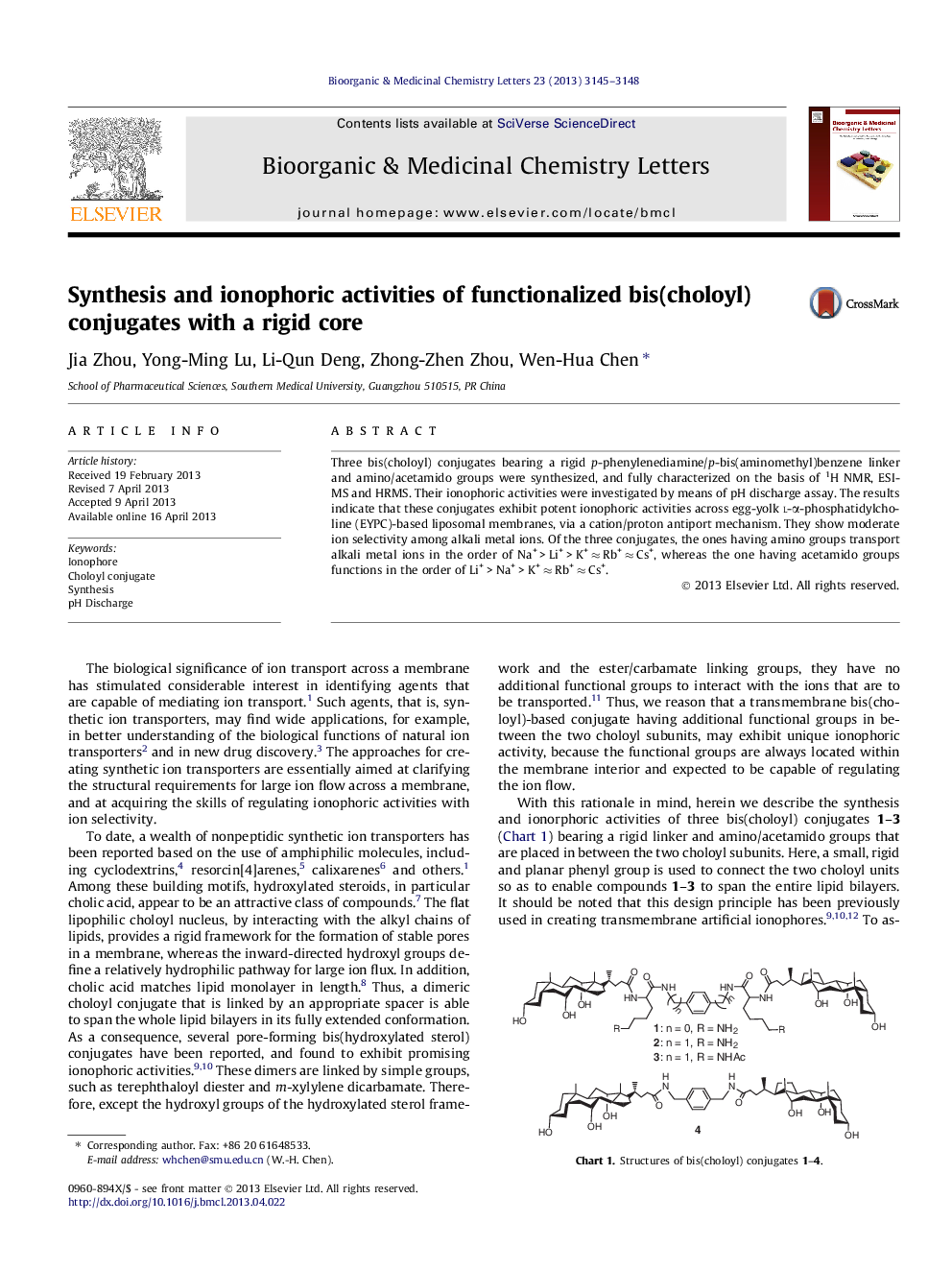
Three bis(choloyl) conjugates bearing a rigid p-phenylenediamine/p-bis(aminomethyl)benzene linker and amino/acetamido groups were synthesized, and fully characterized on the basis of 1H NMR, ESI-MS and HRMS. Their ionophoric activities were investigated by means of pH discharge assay. The results indicate that these conjugates exhibit potent ionophoric activities across egg-yolk l-α-phosphatidylcholine (EYPC)-based liposomal membranes, via a cation/proton antiport mechanism. They show moderate ion selectivity among alkali metal ions. Of the three conjugates, the ones having amino groups transport alkali metal ions in the order of Na+ > Li+ > K+ ≈ Rb+ ≈ Cs+, whereas the one having acetamido groups functions in the order of Li+ > Na+ > K+ ≈ Rb+ ≈ Cs+.
Three functionalized bis(choloyl) conjugates with a rigid core were synthesized, and found to exhibit potent ionophoric activities with ion selectivity across EYPC-based liposomal membranes, via a cation/proton antiport mechanism.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry Letters - Volume 23, Issue 11, 1 June 2013, Pages 3145–3148