| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1372222 | 981867 | 2009 | 4 صفحه PDF | دانلود رایگان |
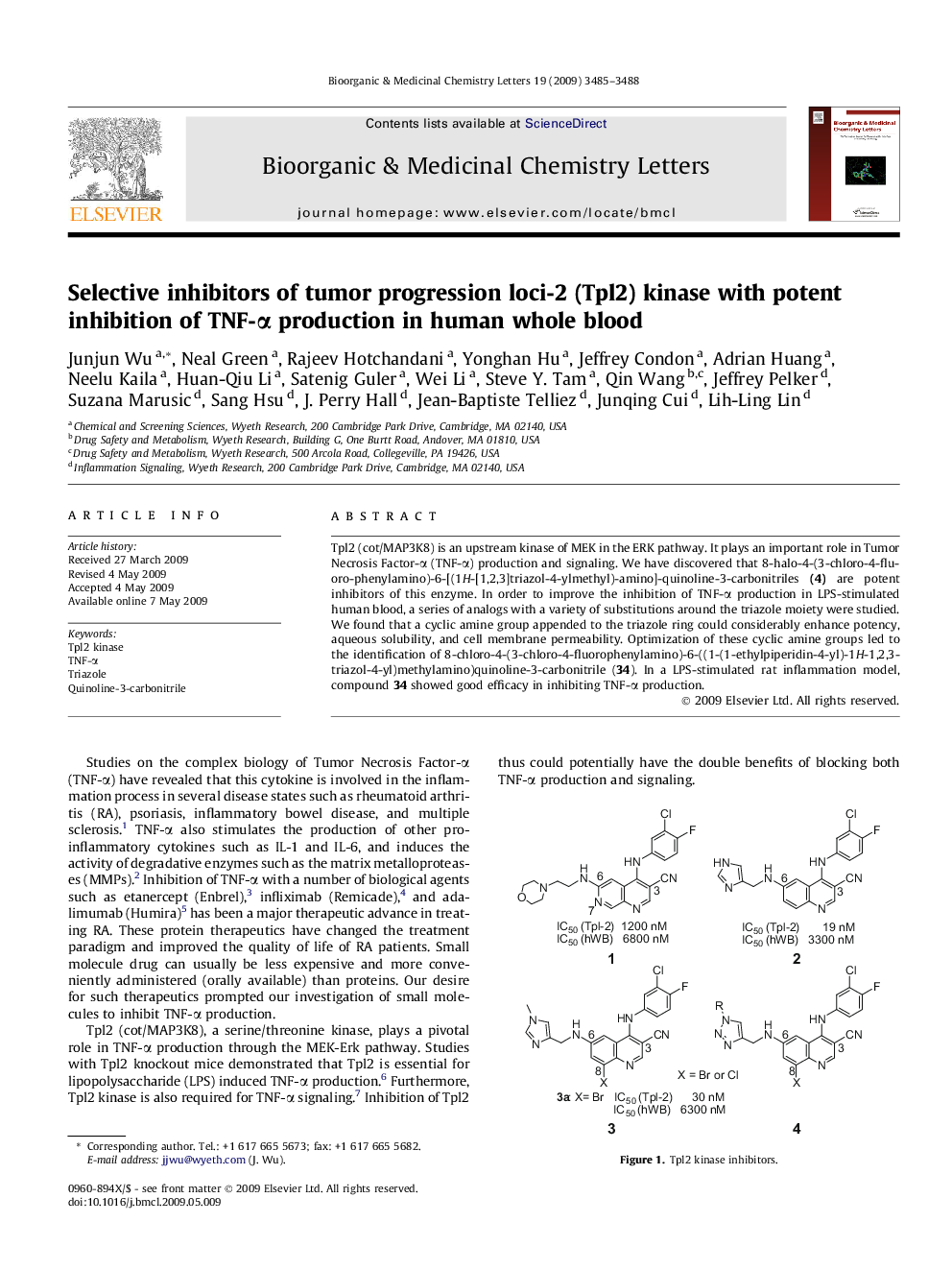
Tpl2 (cot/MAP3K8) is an upstream kinase of MEK in the ERK pathway. It plays an important role in Tumor Necrosis Factor-α (TNF-α) production and signaling. We have discovered that 8-halo-4-(3-chloro-4-fluoro-phenylamino)-6-[(1H-[1,2,3]triazol-4-ylmethyl)-amino]-quinoline-3-carbonitriles (4) are potent inhibitors of this enzyme. In order to improve the inhibition of TNF-α production in LPS-stimulated human blood, a series of analogs with a variety of substitutions around the triazole moiety were studied. We found that a cyclic amine group appended to the triazole ring could considerably enhance potency, aqueous solubility, and cell membrane permeability. Optimization of these cyclic amine groups led to the identification of 8-chloro-4-(3-chloro-4-fluorophenylamino)-6-((1-(1-ethylpiperidin-4-yl)-1H-1,2,3-triazol-4-yl)methylamino)quinoline-3-carbonitrile (34). In a LPS-stimulated rat inflammation model, compound 34 showed good efficacy in inhibiting TNF-α production.
Tpl2 (cot/MAP3K8) is an upstream kinase of MEK in the ERK pathway. It plays an important role in Tumor Necrosis Factor-α (TNF-α) production and signaling. We have discovered that 8-halo-4-(3-chloro-4-fluoro-phenylamino)-6-[(1H-[1,2,3]triazol-4-ylmethyl)-amino]-quinoline-3-carbonitriles (4) are potent inhibitors of this enzyme. In order to improve the inhibition of TNF-α production in LPS-stimulated human blood, a series of analogs with a variety of substitutions around the triazole moiety were studied. We found that a cyclic amine group appended to the triazole ring could considerably enhance potency, aqueous solubility, and cell membrane permeability. Optimization of these cyclic amine groups led to the identification of 8-chloro-4-(3-chloro-4-fluorophenylamino)-6-((1-(1-ethylpiperidin-4-yl)-1H-1,2,3-triazol-4-yl)methylamino)quinoline-3-carbonitrile (34). In a LPS-stimulated rat inflammation model, compound 34 showed good efficacy in inhibiting TNF-α production.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry Letters - Volume 19, Issue 13, 1 July 2009, Pages 3485–3488