| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1375284 | 981936 | 2009 | 5 صفحه PDF | دانلود رایگان |
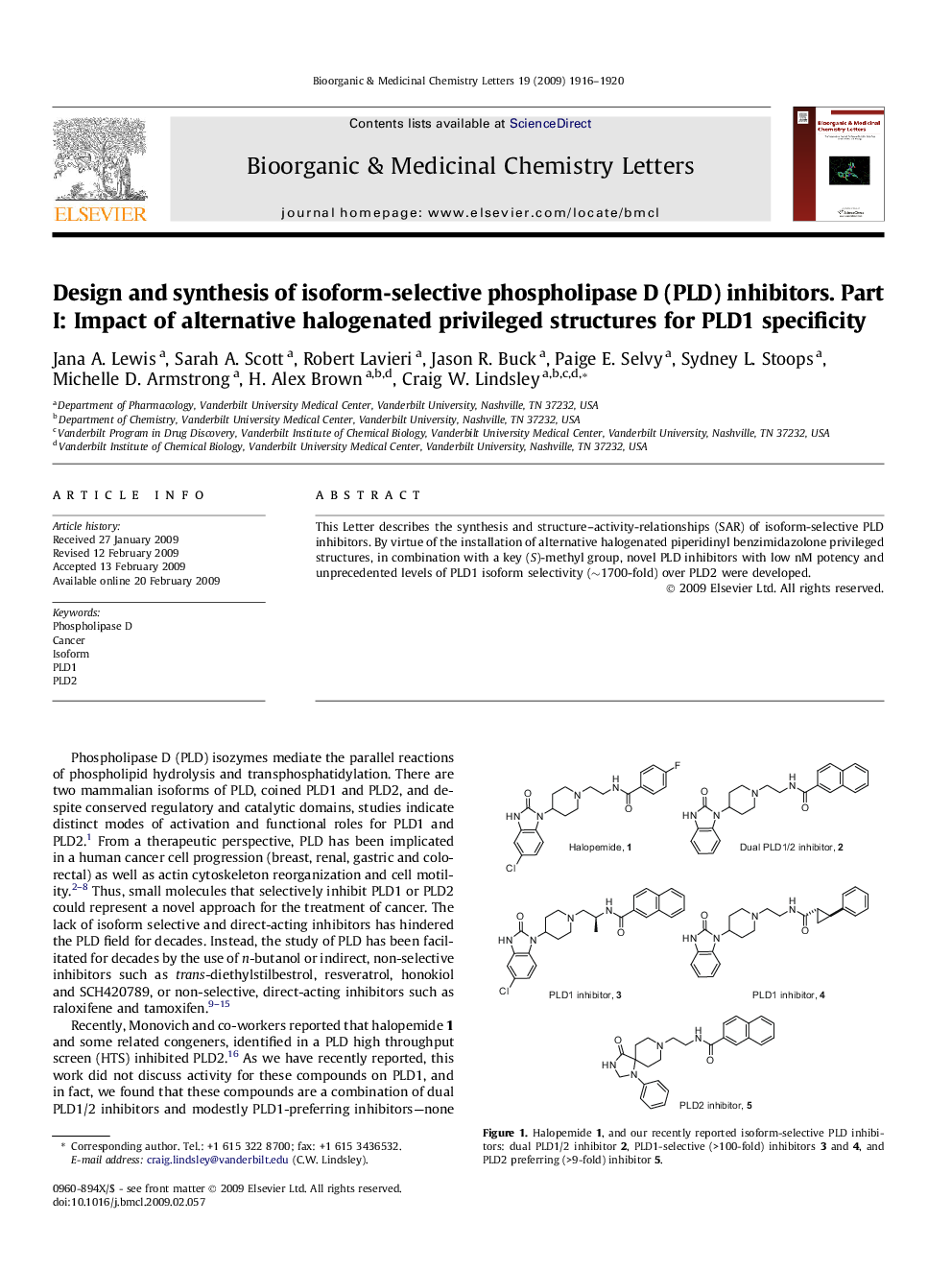
This Letter describes the synthesis and structure–activity-relationships (SAR) of isoform-selective PLD inhibitors. By virtue of the installation of alternative halogenated piperidinyl benzimidazolone privileged structures, in combination with a key (S)-methyl group, novel PLD inhibitors with low nM potency and unprecedented levels of PLD1 isoform selectivity (∼1700-fold) over PLD2 were developed.
The synthesis and SAR of isoform-selective PLD inhibitors is described. By virtue of the installation of alternative halogenated piperidinyl benzimidazolone privileged structures, in combination with a key (S)-methyl group, novel PLD inhibitors with low nM potency and unprecedented levels of isoform selectivity for PLD1 (∼1700-fold) over PLD2 were developed.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry Letters - Volume 19, Issue 7, 1 April 2009, Pages 1916–1920