| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 205534 | 461112 | 2015 | 9 صفحه PDF | دانلود رایگان |
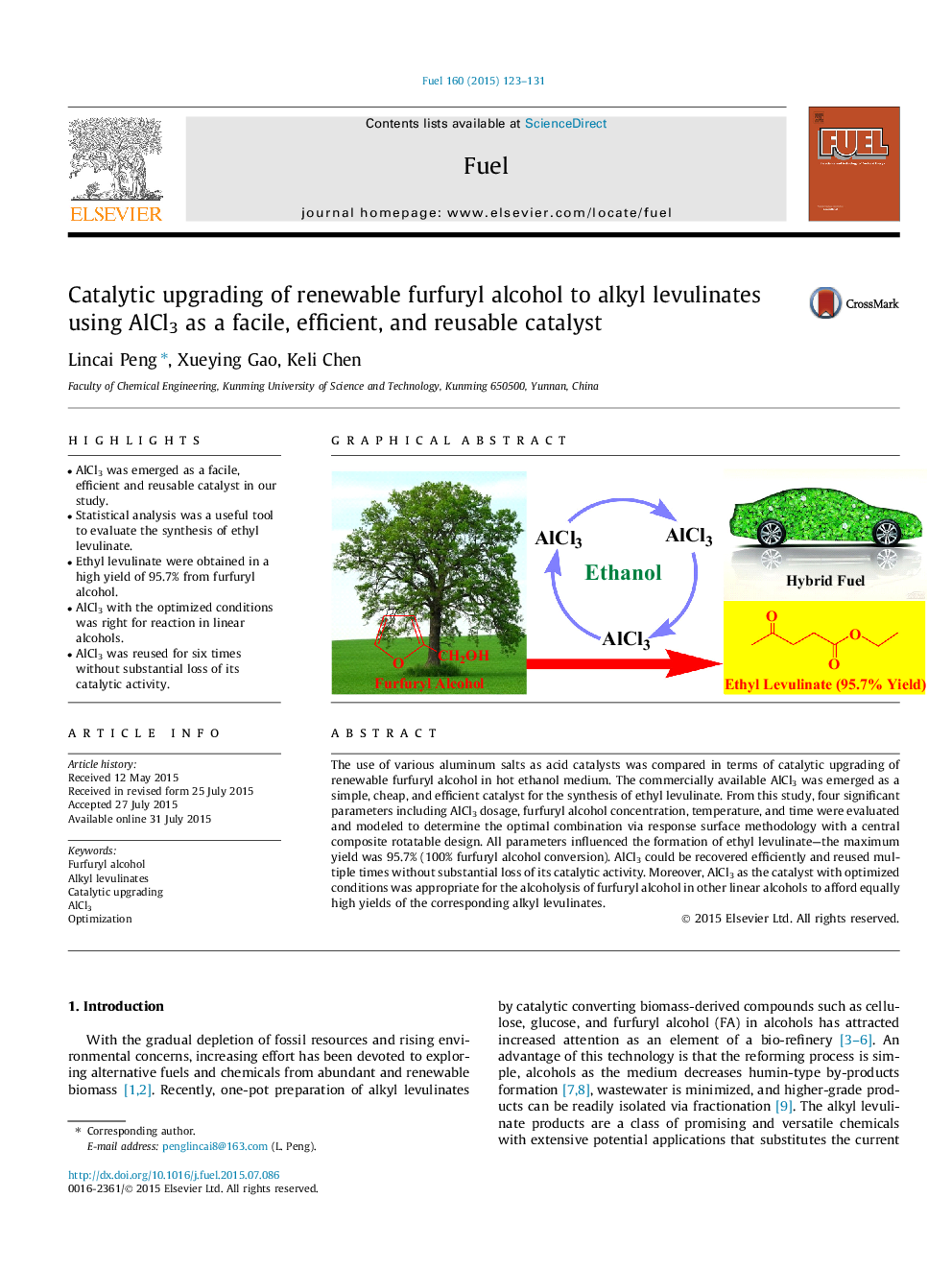
• AlCl3 was emerged as a facile, efficient and reusable catalyst in our study.
• Statistical analysis was a useful tool to evaluate the synthesis of ethyl levulinate.
• Ethyl levulinate were obtained in a high yield of 95.7% from furfuryl alcohol.
• AlCl3 with the optimized conditions was right for reaction in linear alcohols.
• AlCl3 was reused for six times without substantial loss of its catalytic activity.
The use of various aluminum salts as acid catalysts was compared in terms of catalytic upgrading of renewable furfuryl alcohol in hot ethanol medium. The commercially available AlCl3 was emerged as a simple, cheap, and efficient catalyst for the synthesis of ethyl levulinate. From this study, four significant parameters including AlCl3 dosage, furfuryl alcohol concentration, temperature, and time were evaluated and modeled to determine the optimal combination via response surface methodology with a central composite rotatable design. All parameters influenced the formation of ethyl levulinate—the maximum yield was 95.7% (100% furfuryl alcohol conversion). AlCl3 could be recovered efficiently and reused multiple times without substantial loss of its catalytic activity. Moreover, AlCl3 as the catalyst with optimized conditions was appropriate for the alcoholysis of furfuryl alcohol in other linear alcohols to afford equally high yields of the corresponding alkyl levulinates.
Figure optionsDownload as PowerPoint slide
Journal: Fuel - Volume 160, 15 November 2015, Pages 123–131