| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 4371178 | 1302482 | 2013 | 7 صفحه PDF | دانلود رایگان |
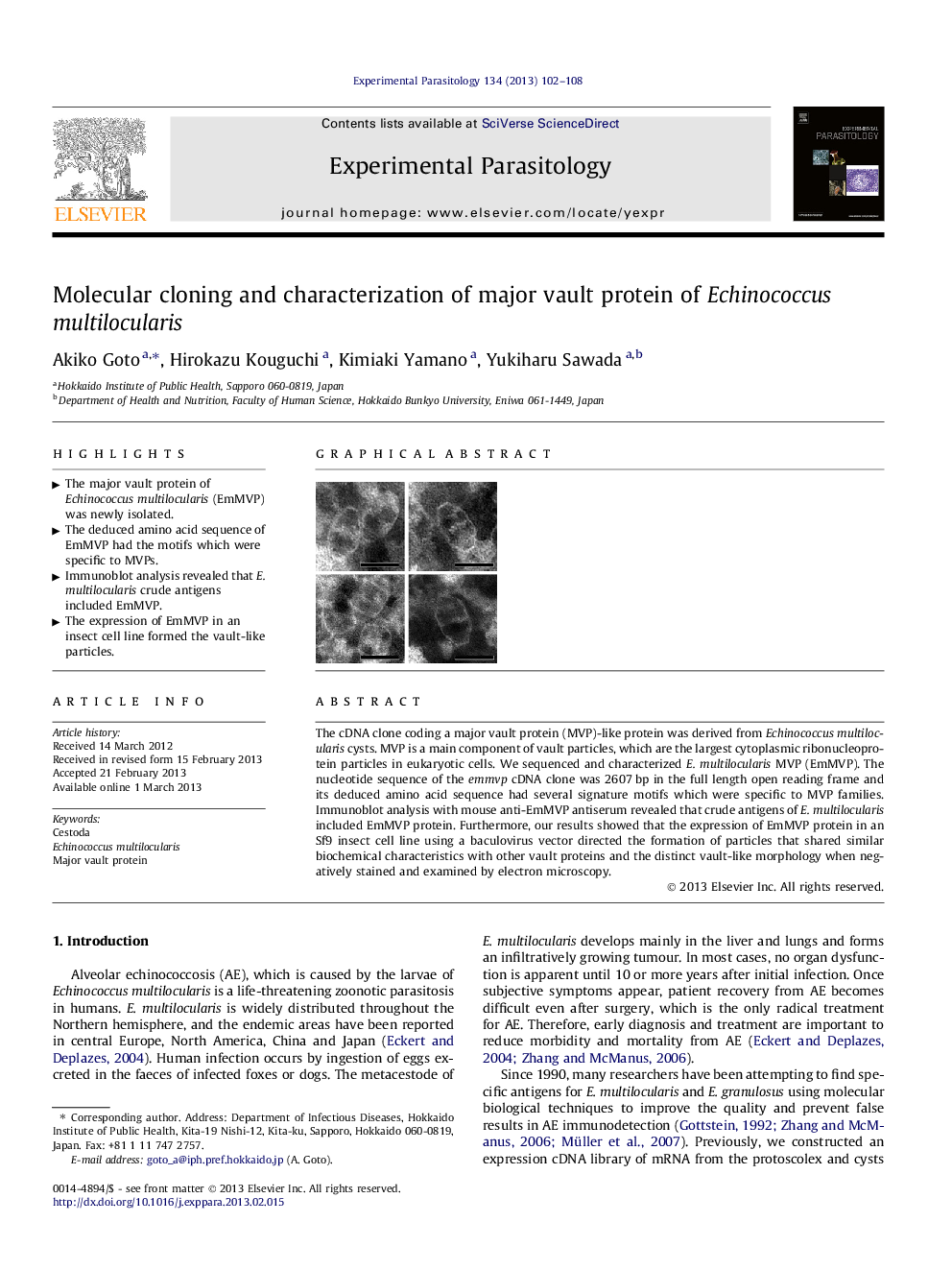
The cDNA clone coding a major vault protein (MVP)-like protein was derived from Echinococcus multilocularis cysts. MVP is a main component of vault particles, which are the largest cytoplasmic ribonucleoprotein particles in eukaryotic cells. We sequenced and characterized E. multilocularis MVP (EmMVP). The nucleotide sequence of the emmvp cDNA clone was 2607 bp in the full length open reading frame and its deduced amino acid sequence had several signature motifs which were specific to MVP families. Immunoblot analysis with mouse anti-EmMVP antiserum revealed that crude antigens of E. multilocularis included EmMVP protein. Furthermore, our results showed that the expression of EmMVP protein in an Sf9 insect cell line using a baculovirus vector directed the formation of particles that shared similar biochemical characteristics with other vault proteins and the distinct vault-like morphology when negatively stained and examined by electron microscopy.
Figure optionsDownload as PowerPoint slideHighlights
► The major vault protein of Echinococcus multilocularis (EmMVP) was newly isolated.
► The deduced amino acid sequence of EmMVP had the motifs which were specific to MVPs.
► Immunoblot analysis revealed that E. multilocularis crude antigens included EmMVP.
► The expression of EmMVP in an insect cell line formed the vault-like particles.
Journal: Experimental Parasitology - Volume 134, Issue 1, May 2013, Pages 102–108