| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 606467 | 1454532 | 2016 | 11 صفحه PDF | دانلود رایگان |
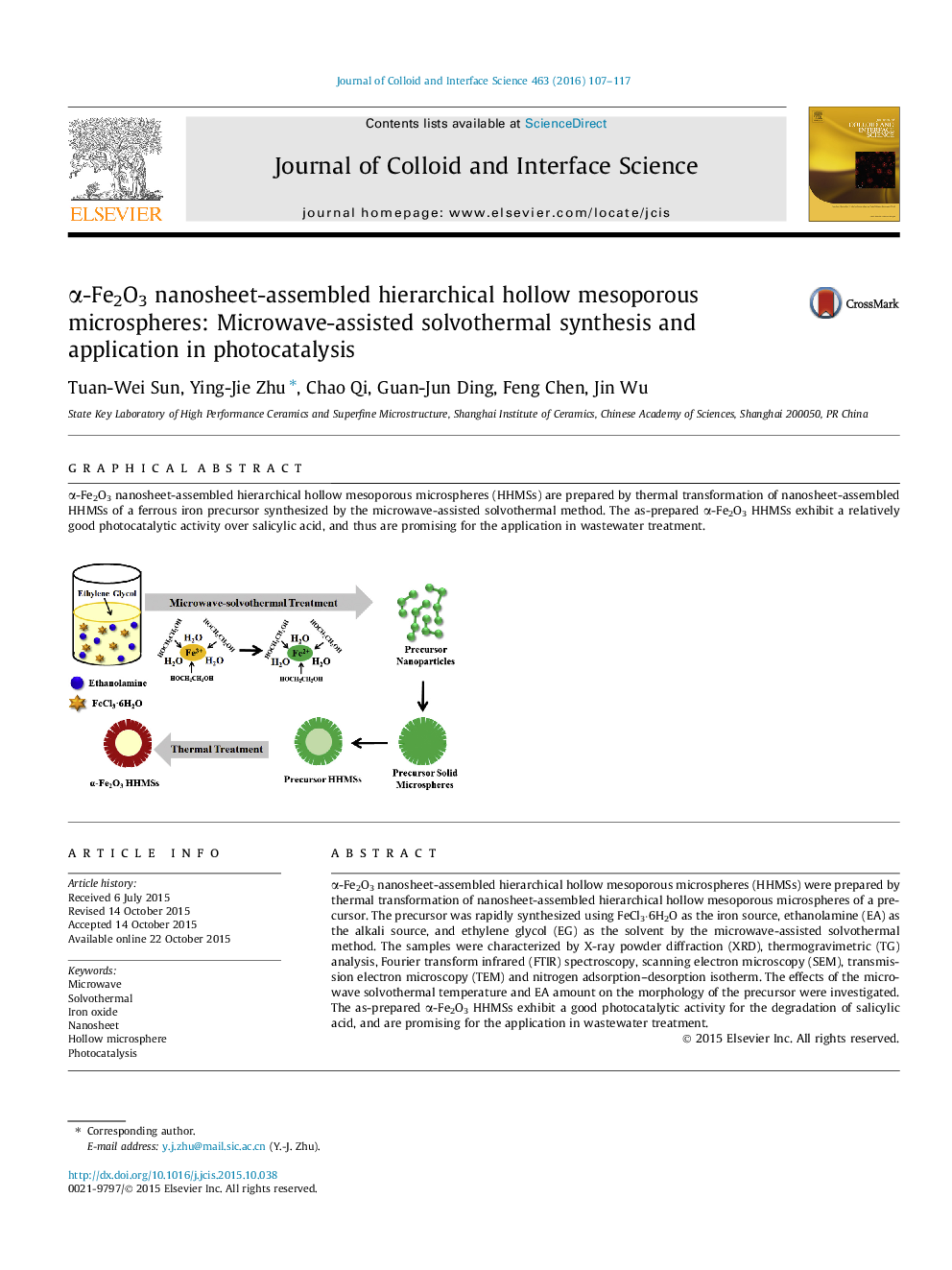
α-Fe2O3 nanosheet-assembled hierarchical hollow mesoporous microspheres (HHMSs) were prepared by thermal transformation of nanosheet-assembled hierarchical hollow mesoporous microspheres of a precursor. The precursor was rapidly synthesized using FeCl3·6H2O as the iron source, ethanolamine (EA) as the alkali source, and ethylene glycol (EG) as the solvent by the microwave-assisted solvothermal method. The samples were characterized by X-ray powder diffraction (XRD), thermogravimetric (TG) analysis, Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy (SEM), transmission electron microscopy (TEM) and nitrogen adsorption–desorption isotherm. The effects of the microwave solvothermal temperature and EA amount on the morphology of the precursor were investigated. The as-prepared α-Fe2O3 HHMSs exhibit a good photocatalytic activity for the degradation of salicylic acid, and are promising for the application in wastewater treatment.
α-Fe2O3 nanosheet-assembled hierarchical hollow mesoporous microspheres (HHMSs) are prepared by thermal transformation of nanosheet-assembled HHMSs of a ferrous iron precursor synthesized by the microwave-assisted solvothermal method. The as-prepared α-Fe2O3 HHMSs exhibit a relatively good photocatalytic activity over salicylic acid, and thus are promising for the application in wastewater treatment.Figure optionsDownload high-quality image (74 K)Download as PowerPoint slide
Journal: Journal of Colloid and Interface Science - Volume 463, 1 February 2016, Pages 107–117