| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 6475345 | 1424969 | 2017 | 13 صفحه PDF | دانلود رایگان |
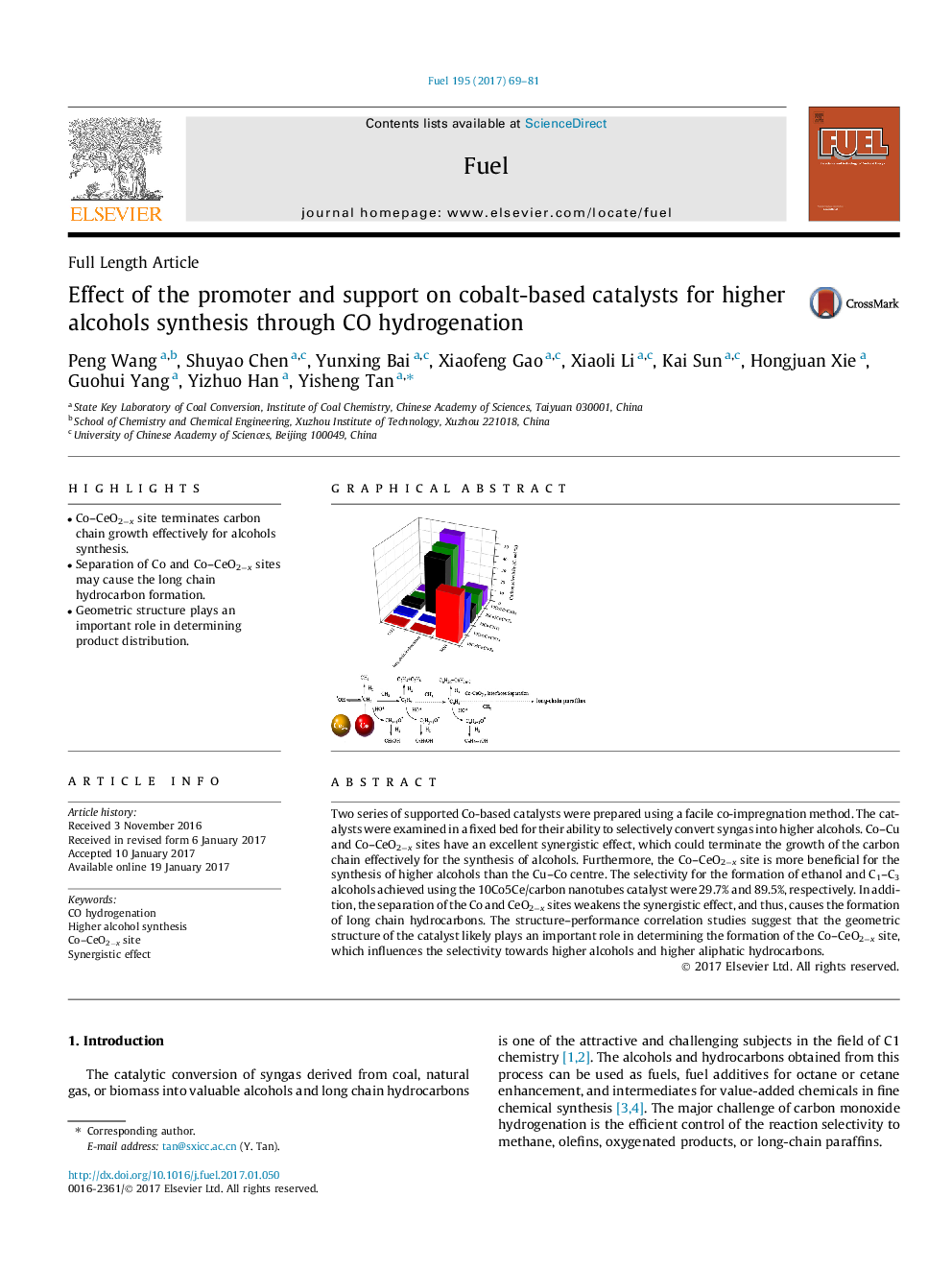
- Co-CeO2âx site terminates carbon chain growth effectively for alcohols synthesis.
- Separation of Co and Co-CeO2âx sites may cause the long chain hydrocarbon formation.
- Geometric structure plays an important role in determining product distribution.
Two series of supported Co-based catalysts were prepared using a facile co-impregnation method. The catalysts were examined in a fixed bed for their ability to selectively convert syngas into higher alcohols. Co-Cu and Co-CeO2âx sites have an excellent synergistic effect, which could terminate the growth of the carbon chain effectively for the synthesis of alcohols. Furthermore, the Co-CeO2âx site is more beneficial for the synthesis of higher alcohols than the Cu-Co centre. The selectivity for the formation of ethanol and C1-C3 alcohols achieved using the 10Co5Ce/carbon nanotubes catalyst were 29.7% and 89.5%, respectively. In addition, the separation of the Co and CeO2âx sites weakens the synergistic effect, and thus, causes the formation of long chain hydrocarbons. The structure-performance correlation studies suggest that the geometric structure of the catalyst likely plays an important role in determining the formation of the Co-CeO2âx site, which influences the selectivity towards higher alcohols and higher aliphatic hydrocarbons.
94
Journal: Fuel - Volume 195, 1 May 2017, Pages 69-81