| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229012 | 1495224 | 2015 | 5 صفحه PDF | دانلود رایگان |
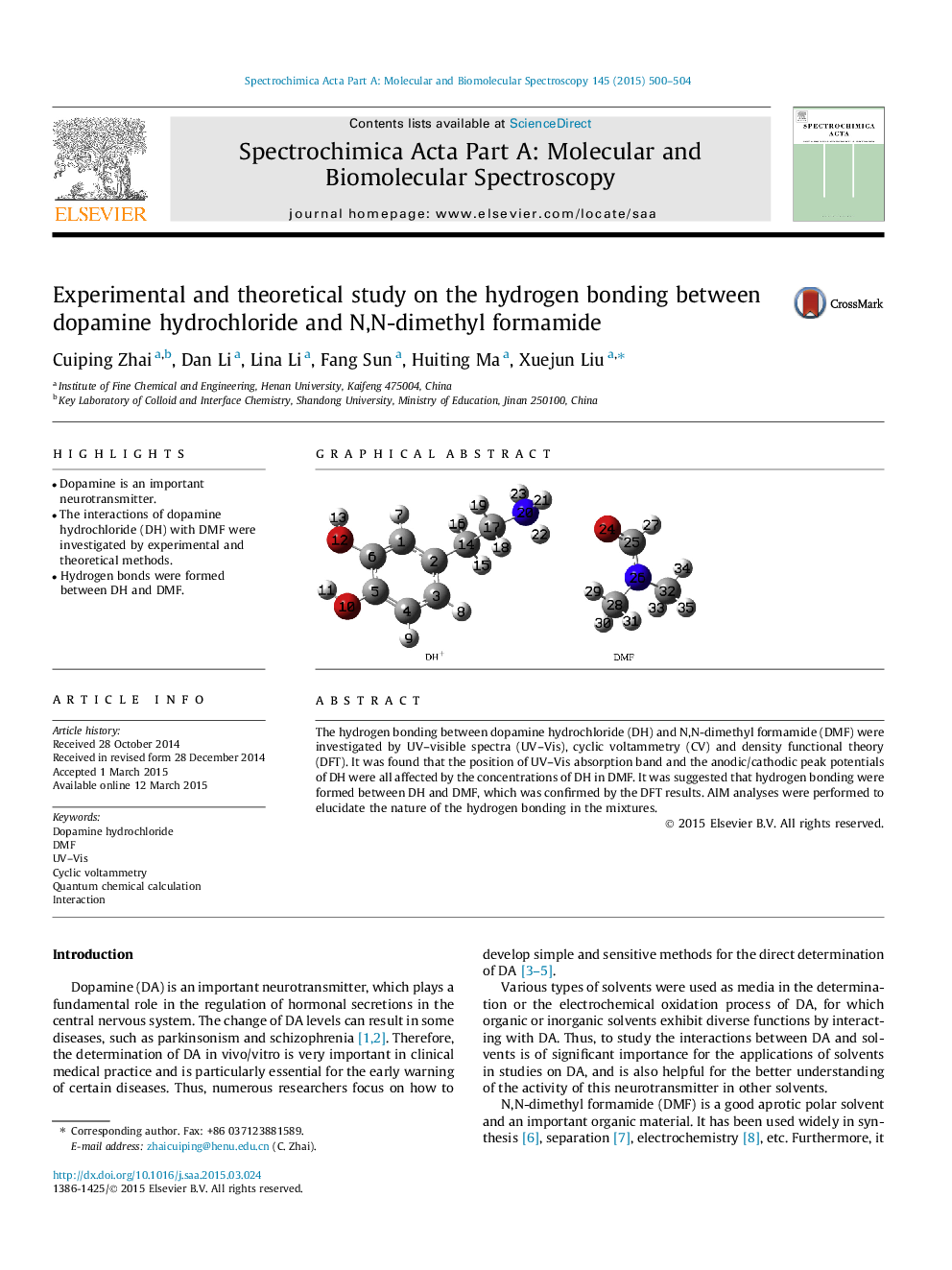
• Dopamine is an important neurotransmitter.
• The interactions of dopamine hydrochloride (DH) with DMF were investigated by experimental and theoretical methods.
• Hydrogen bonds were formed between DH and DMF.
The hydrogen bonding between dopamine hydrochloride (DH) and N,N-dimethyl formamide (DMF) were investigated by UV–visible spectra (UV–Vis), cyclic voltammetry (CV) and density functional theory (DFT). It was found that the position of UV–Vis absorption band and the anodic/cathodic peak potentials of DH were all affected by the concentrations of DH in DMF. It was suggested that hydrogen bonding were formed between DH and DMF, which was confirmed by the DFT results. AIM analyses were performed to elucidate the nature of the hydrogen bonding in the mixtures.
Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 145, 15 June 2015, Pages 500–504