| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1229678 | 1495234 | 2015 | 8 صفحه PDF | دانلود رایگان |
• The binding properties of chelators for Fe(III) is described.
• The pFe values obtained were appreciably higher than deferiprone.
• Ligands binds by imine nitrogens at low pH and by phenolic oxygens at high pH.
• Ligands are forming strong tris(iminophenolate) type of complex.
• Both the ligands can be employed for iron(III) intoxication.
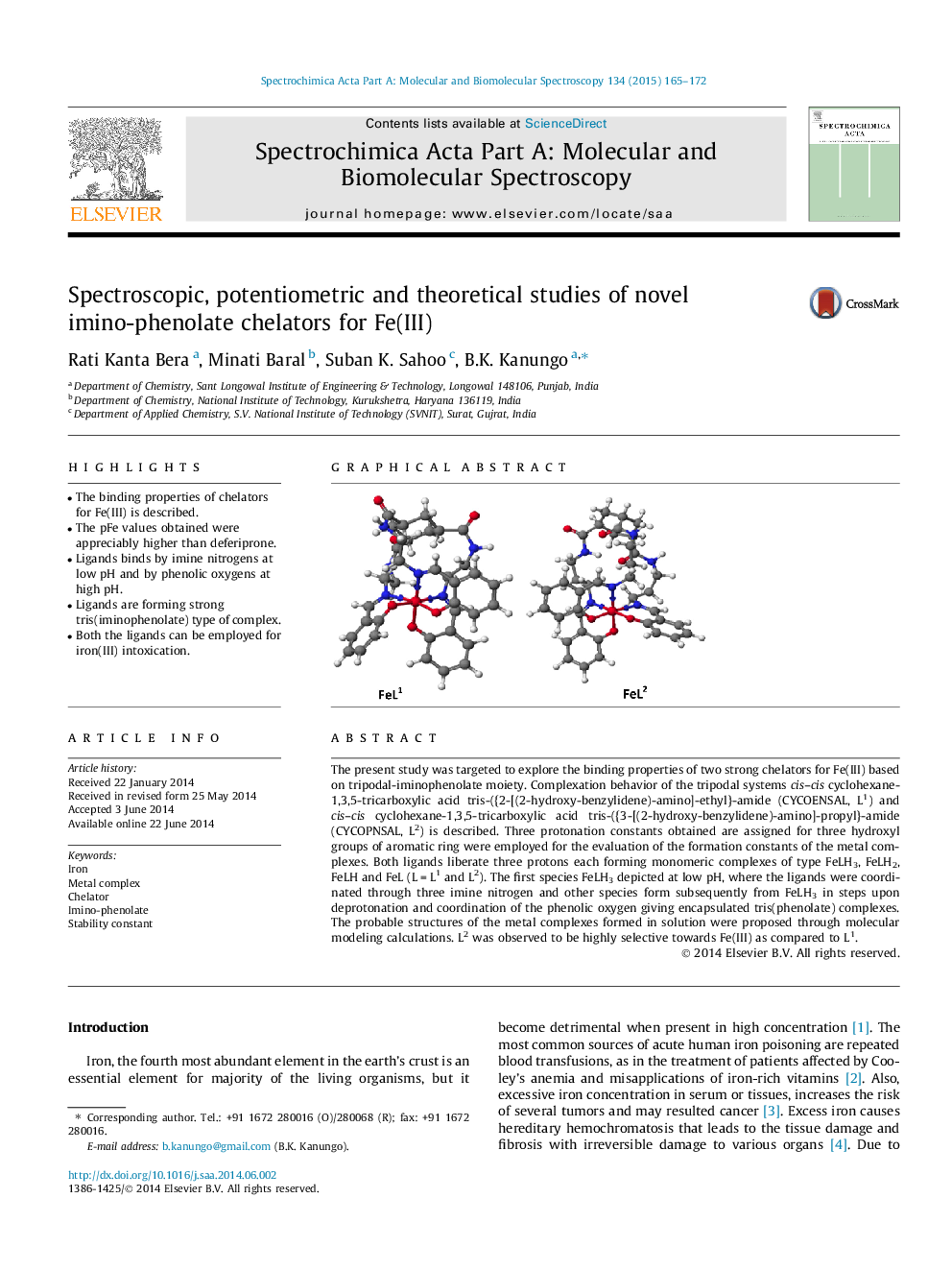
The present study was targeted to explore the binding properties of two strong chelators for Fe(III) based on tripodal-iminophenolate moiety. Complexation behavior of the tripodal systems cis–cis cyclohexane-1,3,5-tricarboxylic acid tris-({2-[(2-hydroxy-benzylidene)-amino]-ethyl}-amide (CYCOENSAL, L1) and cis–cis cyclohexane-1,3,5-tricarboxylic acid tris-({3-[(2-hydroxy-benzylidene)-amino]-propyl}-amide (CYCOPNSAL, L2) is described. Three protonation constants obtained are assigned for three hydroxyl groups of aromatic ring were employed for the evaluation of the formation constants of the metal complexes. Both ligands liberate three protons each forming monomeric complexes of type FeLH3, FeLH2, FeLH and FeL (L = L1 and L2). The first species FeLH3 depicted at low pH, where the ligands were coordinated through three imine nitrogen and other species form subsequently from FeLH3 in steps upon deprotonation and coordination of the phenolic oxygen giving encapsulated tris(phenolate) complexes. The probable structures of the metal complexes formed in solution were proposed through molecular modeling calculations. L2 was observed to be highly selective towards Fe(III) as compared to L1.
Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 134, 5 January 2015, Pages 165–172