| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1234006 | 1495241 | 2014 | 9 صفحه PDF | دانلود رایگان |
• Vibrational spectroscopic investigations of cyanuric chloride were carried out.
• NBO analysis used to explain the interaction between electron donors and acceptors.
• HOMO and LUMO energy gap explains the eventual charge transfer interactions.
• Thermodynamic parameters of the title molecule are reported.
The vibrational spectral analysis of cyanuric chloride was carried out by using FT-Raman and FT-IR spectra in the range 100–4000 cm−1 and 400–4000 cm−1 respectively. The structure optimization was done and structural characteristics were determined by Density Functional Theory (B3LYP) method with 6-31G(d,p) and 6-311++G(d,p) basis sets. The vibrational wavenumbers have been calculated and scaled values are compared with experimental FT-IR and FT-Raman spectra. A detailed interpretation of the vibrational spectra of the molecule has been made on the basis of the calculated Potential Energy Distribution (PED). The Natural bonding orbital (NBO) analysis performed to confirm the stability of the molecule arising from hyper conjugation and delocalization. The Mulliken atomic charges were also calculated. The computed HOMO–LUMO energy gap shows that charge transfer occurs within the molecule. The thermodynamic properties at different temperatures have been calculated from the vibrational analysis.
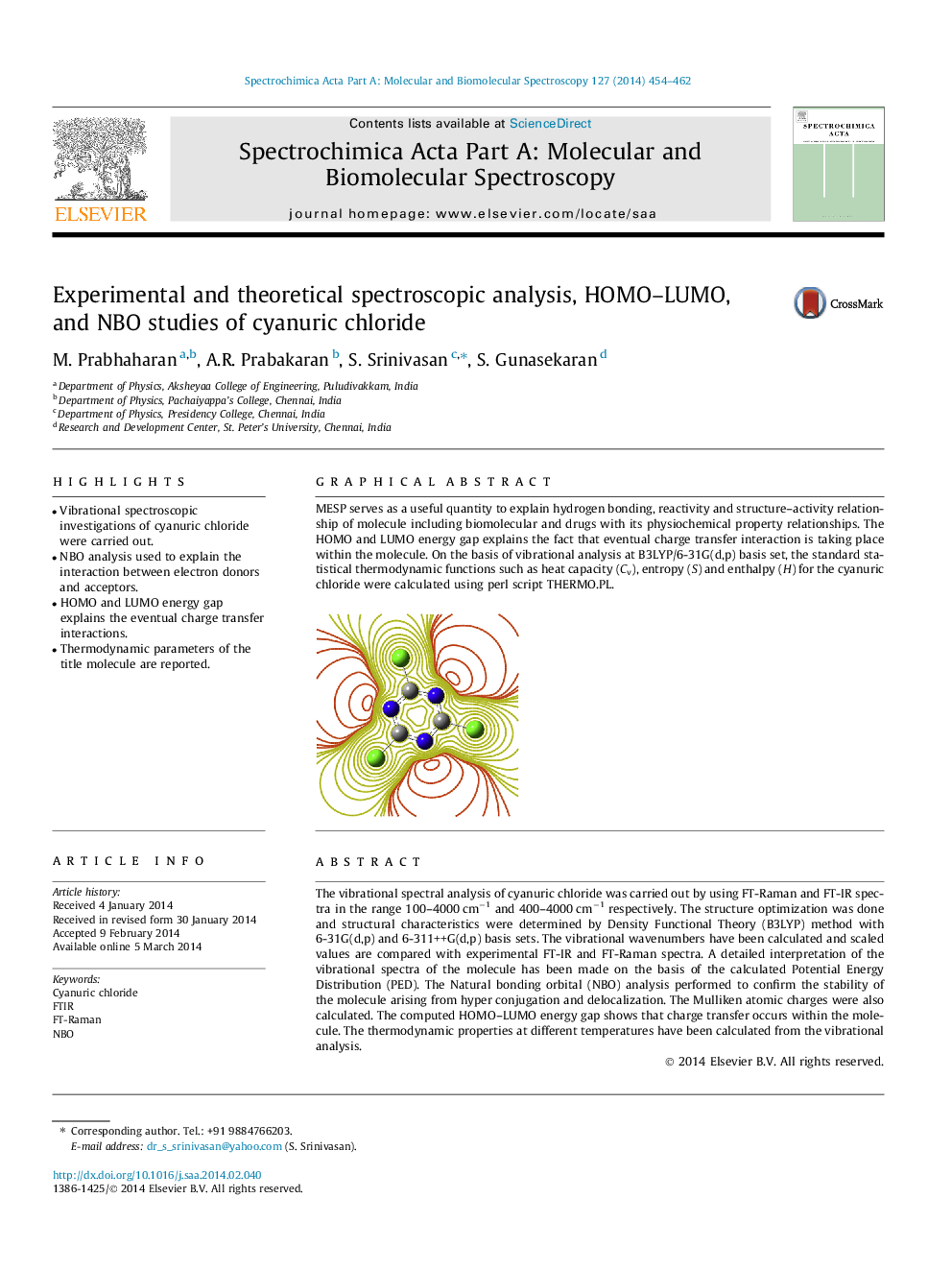
MESP serves as a useful quantity to explain hydrogen bonding, reactivity and structure–activity relationship of molecule including biomolecular and drugs with its physiochemical property relationships. The HOMO and LUMO energy gap explains the fact that eventual charge transfer interaction is taking place within the molecule. On the basis of vibrational analysis at B3LYP/6-31G(d,p) basis set, the standard statistical thermodynamic functions such as heat capacity (Cv), entropy (S) and enthalpy (H) for the cyanuric chloride were calculated using perl script THERMO.PL.Figure optionsDownload as PowerPoint slide
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 127, 5 June 2014, Pages 454–462