| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1238205 | 968922 | 2006 | 6 صفحه PDF | دانلود رایگان |
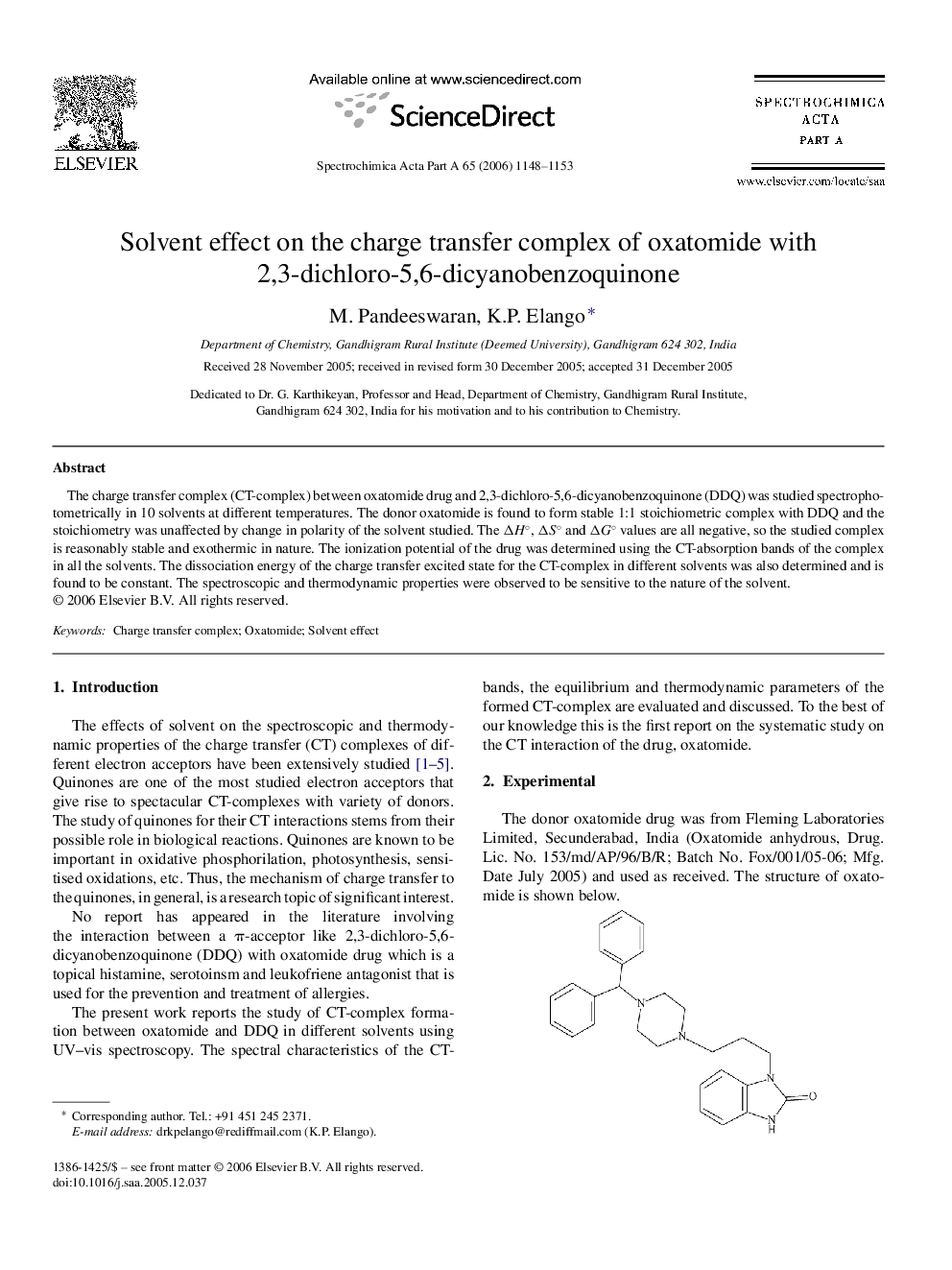
The charge transfer complex (CT-complex) between oxatomide drug and 2,3-dichloro-5,6-dicyanobenzoquinone (DDQ) was studied spectrophotometrically in 10 solvents at different temperatures. The donor oxatomide is found to form stable 1:1 stoichiometric complex with DDQ and the stoichiometry was unaffected by change in polarity of the solvent studied. The ΔH°, ΔS° and ΔG° values are all negative, so the studied complex is reasonably stable and exothermic in nature. The ionization potential of the drug was determined using the CT-absorption bands of the complex in all the solvents. The dissociation energy of the charge transfer excited state for the CT-complex in different solvents was also determined and is found to be constant. The spectroscopic and thermodynamic properties were observed to be sensitive to the nature of the solvent.
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 65, Issue 5, December 2006, Pages 1148–1153