| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1306542 | 975094 | 2011 | 7 صفحه PDF | دانلود رایگان |
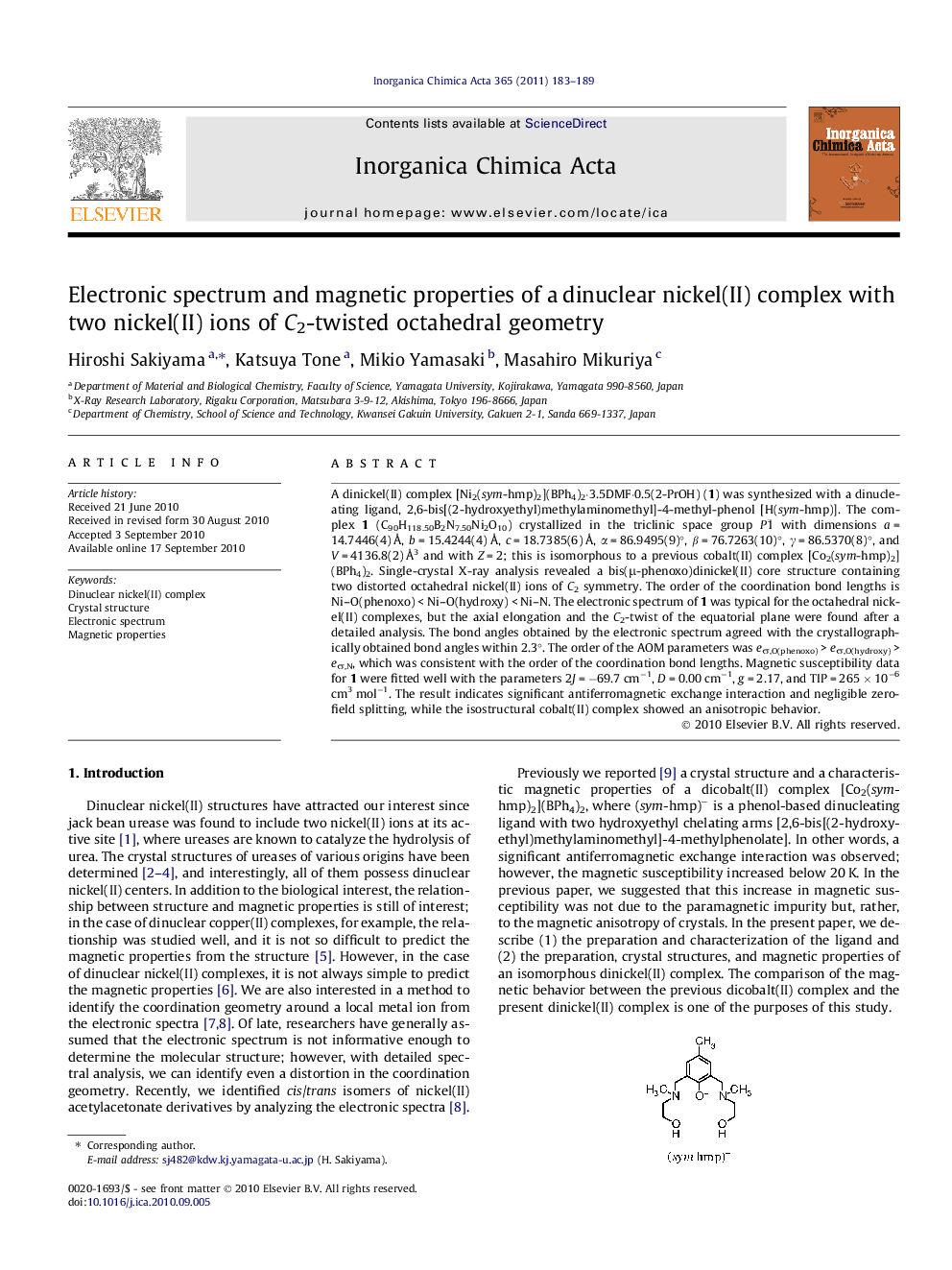
A dinickel(II) complex [Ni2(sym-hmp)2](BPh4)2·3.5DMF·0.5(2-PrOH) (1) was synthesized with a dinucleating ligand, 2,6-bis[(2-hydroxyethyl)methylaminomethyl]-4-methyl-phenol [H(sym-hmp)]. The complex 1 (C90H118.50B2N7.50Ni2O10) crystallized in the triclinic space group P1¯ with dimensions a = 14.7446(4) Å, b = 15.4244(4) Å, c = 18.7385(6) Å, α = 86.9495(9)°, β = 76.7263(10)°, γ = 86.5370(8)°, and V = 4136.8(2) Å3 and with Z = 2; this is isomorphous to a previous cobalt(II) complex [Co2(sym-hmp)2](BPh4)2. Single-crystal X-ray analysis revealed a bis(μ-phenoxo)dinickel(II) core structure containing two distorted octahedral nickel(II) ions of C2 symmetry. The order of the coordination bond lengths is Ni–O(phenoxo) < Ni–O(hydroxy) < Ni–N. The electronic spectrum of 1 was typical for the octahedral nickel(II) complexes, but the axial elongation and the C2-twist of the equatorial plane were found after a detailed analysis. The bond angles obtained by the electronic spectrum agreed with the crystallographically obtained bond angles within 2.3°. The order of the AOM parameters was eσ,O(phenoxo) > eσ,O(hydroxy) > eσ,N, which was consistent with the order of the coordination bond lengths. Magnetic susceptibility data for 1 were fitted well with the parameters 2J = −69.7 cm−1, D = 0.00 cm−1, g = 2.17, and TIP = 265 × 10−6 cm3 mol−1. The result indicates significant antiferromagnetic exchange interaction and negligible zero-field splitting, while the isostructural cobalt(II) complex showed an anisotropic behavior.
The electronic spectrum of a dinickel(II) complex [Ni2(sym-hmp)2](BPh4)2·3.5DMF·0.5(2-PrOH) [H(sym-hmp) = 2,6-bis[(2-hydroxyethyl)methylaminomethyl]-4-methyl-phenol] was analyzed on the basis of the angular overlap model (AOM), and the bond angles obtained by the electronic spectrum agreed with the crystallographically obtained bond angles within 2.3°. The order of the AOM parameters was eσ,O(phenoxo) > eσ,O(hydroxy) > eσ,N, which was consistent with the order of the coordination bond lengths.Figure optionsDownload as PowerPoint slide
Journal: Inorganica Chimica Acta - Volume 365, Issue 1, 15 January 2011, Pages 183–189