| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1308471 | 975174 | 2009 | 6 صفحه PDF | دانلود رایگان |
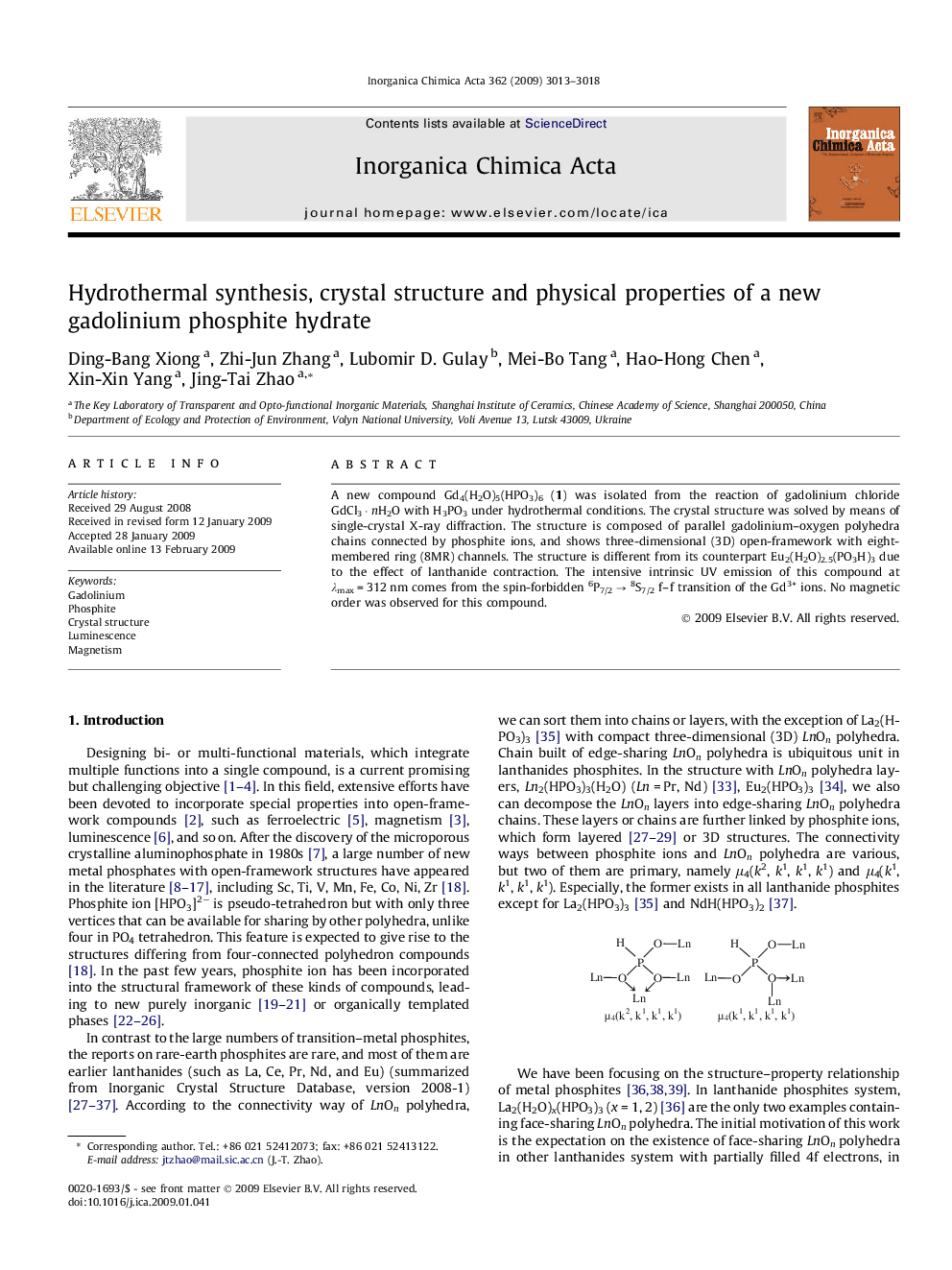
A new compound Gd4(H2O)5(HPO3)6 (1) was isolated from the reaction of gadolinium chloride GdCl3 · nH2O with H3PO3 under hydrothermal conditions. The crystal structure was solved by means of single-crystal X-ray diffraction. The structure is composed of parallel gadolinium–oxygen polyhedra chains connected by phosphite ions, and shows three-dimensional (3D) open-framework with eight-membered ring (8MR) channels. The structure is different from its counterpart Eu2(H2O)2.5(PO3H)3 due to the effect of lanthanide contraction. The intensive intrinsic UV emission of this compound at λmax = 312 nm comes from the spin-forbidden 6P7/2 → 8S7/2 f–f transition of the Gd3+ ions. No magnetic order was observed for this compound.
A new compound Gd4(H2O)5(HPO3)6 was obtained by hydrothermal methods. The crystal structure is built up of parallel gadolinium–oxygen polyhedra chains connected into three-dimensional (3D) open-framework by phosphite ions, showing eight-membered ring channels. The compound shows intrinsic intensive UV emission around 312 nm and no magnetic order was observed.Figure optionsDownload as PowerPoint slide
Journal: Inorganica Chimica Acta - Volume 362, Issue 9, 1 July 2009, Pages 3013–3018