| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1310803 | 1499198 | 2012 | 8 صفحه PDF | دانلود رایگان |
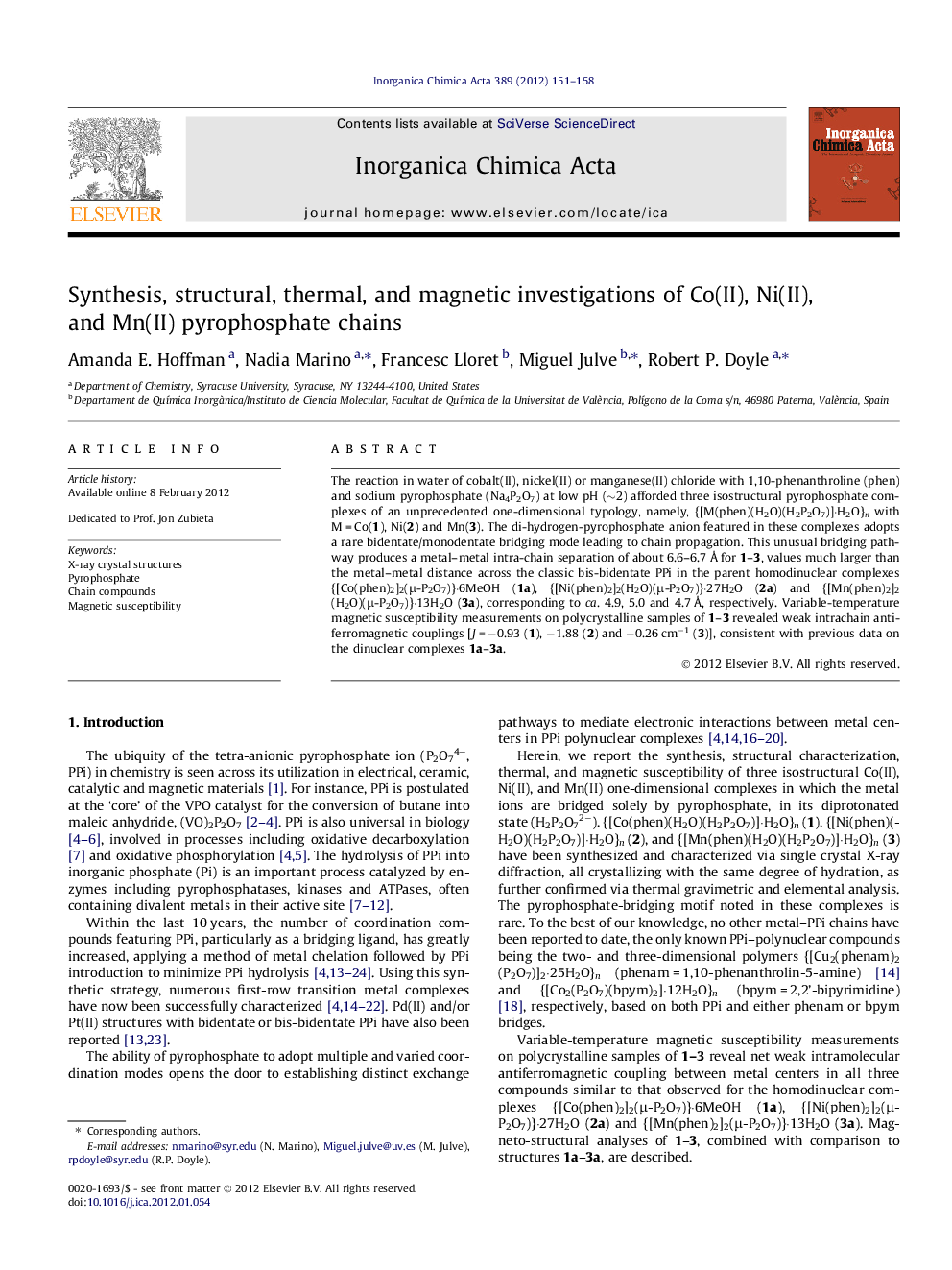
The reaction in water of cobalt(II), nickel(II) or manganese(II) chloride with 1,10-phenanthroline (phen) and sodium pyrophosphate (Na4P2O7) at low pH (∼2) afforded three isostructural pyrophosphate complexes of an unprecedented one-dimensional typology, namely, {[M(phen)(H2O)(H2P2O7)]·H2O}n with M = Co(1), Ni(2) and Mn(3). The di-hydrogen-pyrophosphate anion featured in these complexes adopts a rare bidentate/monodentate bridging mode leading to chain propagation. This unusual bridging pathway produces a metal–metal intra-chain separation of about 6.6–6.7 Å for 1–3, values much larger than the metal–metal distance across the classic bis-bidentate PPi in the parent homodinuclear complexes {[Co(phen)2]2(μ-P2O7)}·6MeOH (1a), {[Ni(phen)2]2(H2O)(μ-P2O7)}·27H2O (2a) and {[Mn(phen)2]2(H2O)(μ-P2O7)}·13H2O (3a), corresponding to ca. 4.9, 5.0 and 4.7 Å, respectively. Variable-temperature magnetic susceptibility measurements on polycrystalline samples of 1–3 revealed weak intrachain antiferromagnetic couplings [J = −0.93 (1), −1.88 (2) and −0.26 cm−1 (3)], consistent with previous data on the dinuclear complexes 1a–3a.
(I) Perspective view of a fragment of the neutral {[Ni(phen)(H2O)(H2P2O7)]·H2O}n chain [the corresponding Mn(II) and Co(II) derivatives are isostructural]; (II) PPi (H2P2O72−) coordination mode and typology in (a) {[Cu(bipy)(cis-H2P2O7)]2}·3H2O and {[Cu(bipy)(trans-H2P2O7)]2} and (b) the polymeric chains shown in (I).Figure optionsDownload as PowerPoint slideHighlights
► Isostructural, dihydrogenpyrophosphate-bridged complexes of Co(II), Ni(II) or Mn(II).
► The synthesis of these complexes was attainable only through careful manipulation of pH.
► Magnetic coupling through the bidentate/monodentate pathway is antiferromagnetic.
Journal: Inorganica Chimica Acta - Volume 389, 1 July 2012, Pages 151–158