| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1322661 | 977229 | 2010 | 6 صفحه PDF | دانلود رایگان |
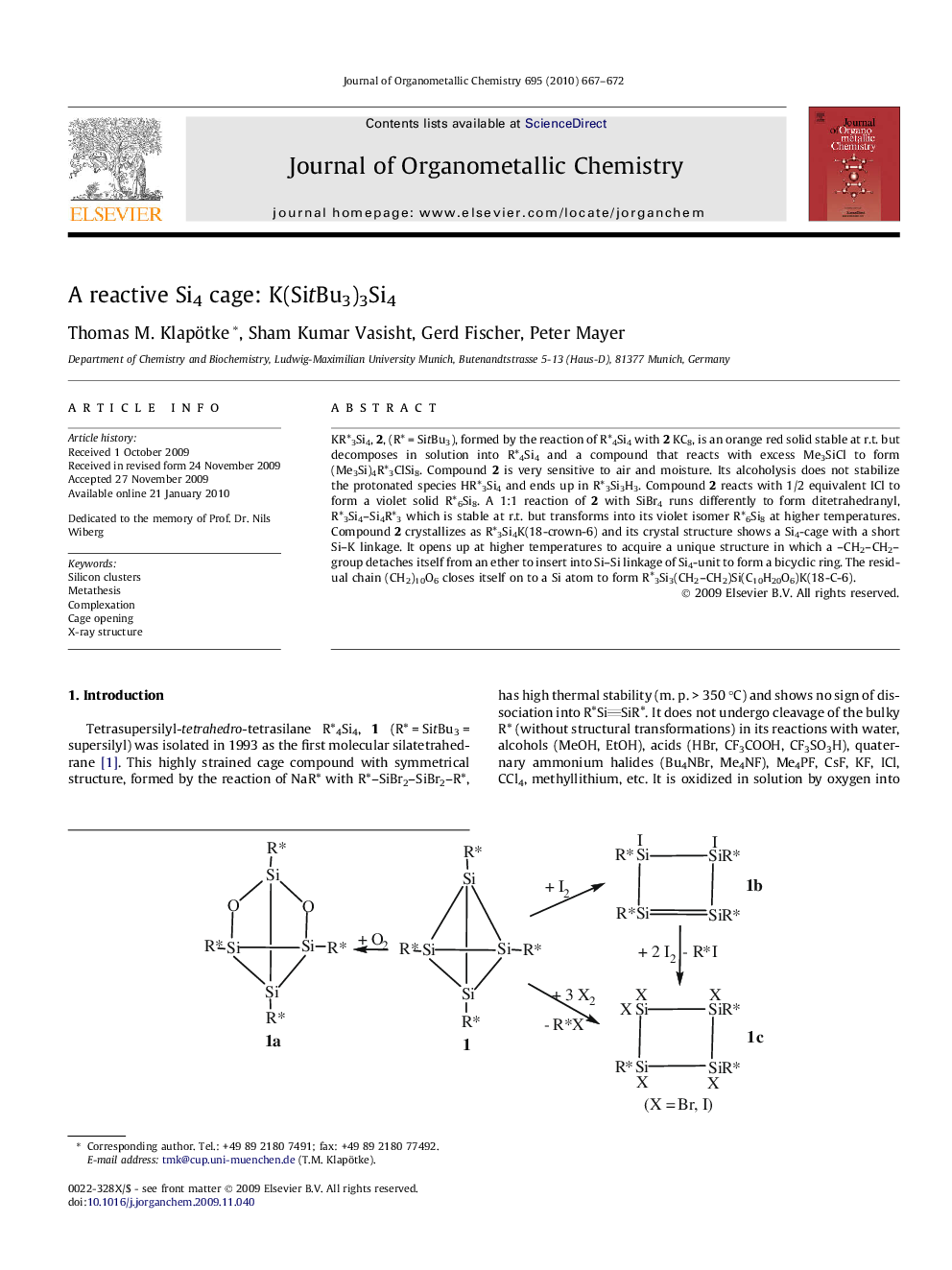
KR∗3Si4, 2, (R∗ = SitBu3), formed by the reaction of R∗4Si4 with 2 KC8, is an orange red solid stable at r.t. but decomposes in solution into R∗4Si4 and a compound that reacts with excess Me3SiCl to form (Me3Si)4R∗3ClSi8. Compound 2 is very sensitive to air and moisture. Its alcoholysis does not stabilize the protonated species HR∗3Si4 and ends up in R∗3Si3H3. Compound 2 reacts with 1/2 equivalent ICl to form a violet solid R∗6Si8. A 1:1 reaction of 2 with SiBr4 runs differently to form ditetrahedranyl, R∗3Si4–Si4R∗3 which is stable at r.t. but transforms into its violet isomer R∗6Si8 at higher temperatures. Compound 2 crystallizes as R∗3Si4K(18-crown-6) and its crystal structure shows a Si4-cage with a short Si–K linkage. It opens up at higher temperatures to acquire a unique structure in which a –CH2–CH2– group detaches itself from an ether to insert into Si–Si linkage of Si4-unit to form a bicyclic ring. The residual chain (CH2)10O6 closes itself on to a Si atom to form R∗3Si3(CH2–CH2)Si(C10H20O6)K(18-C-6).
KR∗3Si4, (R∗ = SitBu3), formed by the reaction of R∗4Si4 with 2 KC8, is a orange red solid which complexes with 18-crown-6 to form a red solid, R∗3Si4K(18-crown-6) A.Figure optionsDownload as PowerPoint slide
Journal: Journal of Organometallic Chemistry - Volume 695, Issue 5, 1 March 2010, Pages 667–672