| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1328346 | 1499949 | 2007 | 10 صفحه PDF | دانلود رایگان |
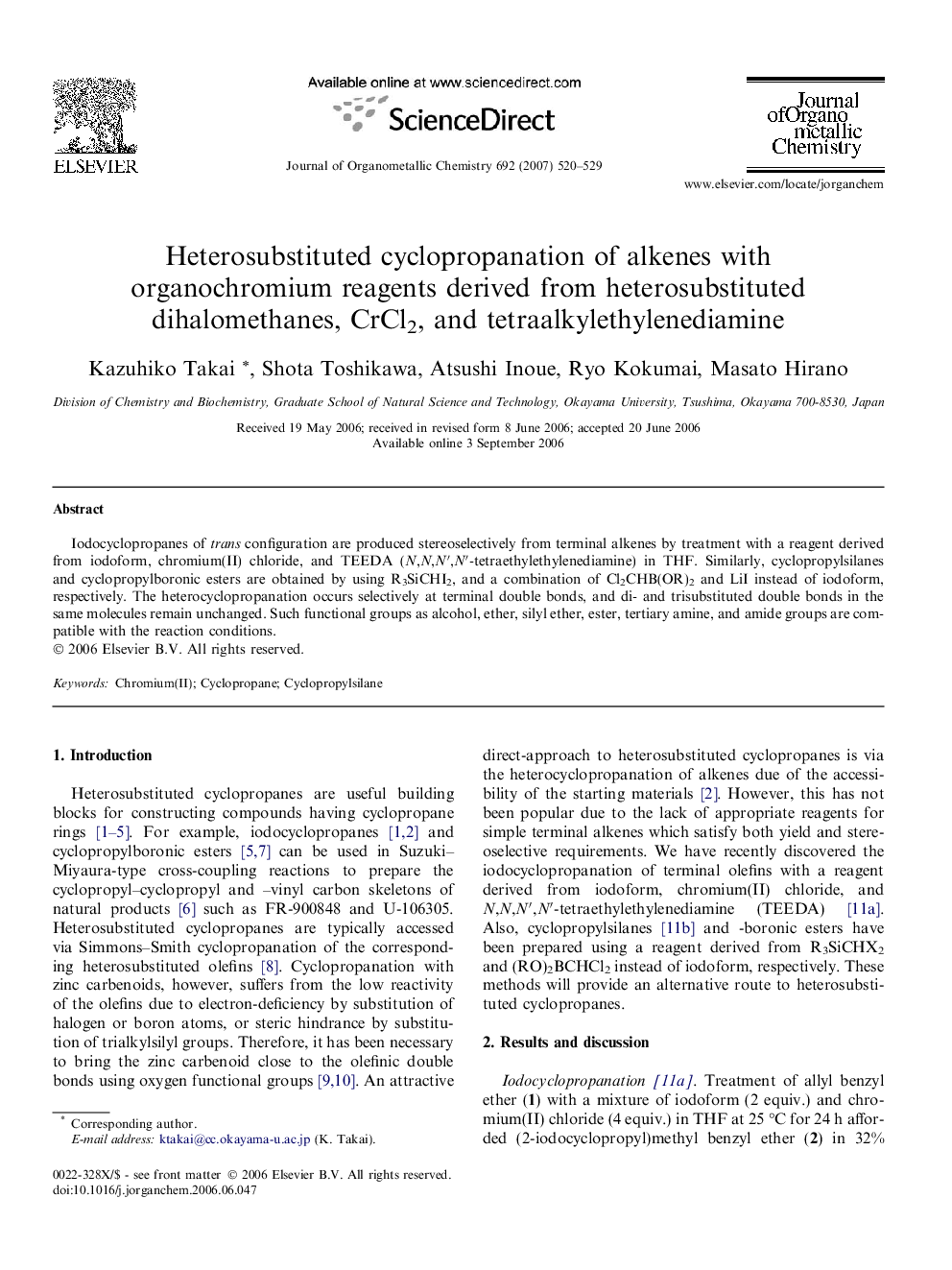
Iodocyclopropanes of trans configuration are produced stereoselectively from terminal alkenes by treatment with a reagent derived from iodoform, chromium(II) chloride, and TEEDA (N,N,N′,N′-tetraethylethylenediamine) in THF. Similarly, cyclopropylsilanes and cyclopropylboronic esters are obtained by using R3SiCHI2, and a combination of Cl2CHB(OR)2 and LiI instead of iodoform, respectively. The heterocyclopropanation occurs selectively at terminal double bonds, and di- and trisubstituted double bonds in the same molecules remain unchanged. Such functional groups as alcohol, ether, silyl ether, ester, tertiary amine, and amide groups are compatible with the reaction conditions.
Iodocyclopropanes of trans configuration are produced stereoselectively from terminal alkenes by treatment with a reagent derived from iodoform, chromium(II) chloride, and N,N,N′,N′-tetraethylethylenediamine in THF. Similarly, cyclopropylsilanes and -boronic esters are obtained by using R3SiCHI2, and a combination of Cl2CHB(OR)2 and LiI instead of iodoform, respectively.Figure optionsDownload as PowerPoint slide
Journal: Journal of Organometallic Chemistry - Volume 692, Issues 1–3, 1 January 2007, Pages 520–529