| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1358964 | 981374 | 2011 | 7 صفحه PDF | دانلود رایگان |
A series of thiazolidine-4-carboxylic acid derivatives were synthesized and evaluated for their ability to inhibit neuraminidase (NA) of influenza A virus. All the compounds were synthesized in good yields starting from commercially available l-cysteine hydrochloride using a suitable synthetic strategy. These compounds showed moderate inhibitory activity against influenza A neuraminidase. The most potent compound of this series is compound 4f (IC50 = 0.14 μM), which is about sevenfold less potent than oseltamivir and could be used to design novel influenza NA inhibitors that exhibit increased activity based on thiazolidine ring.
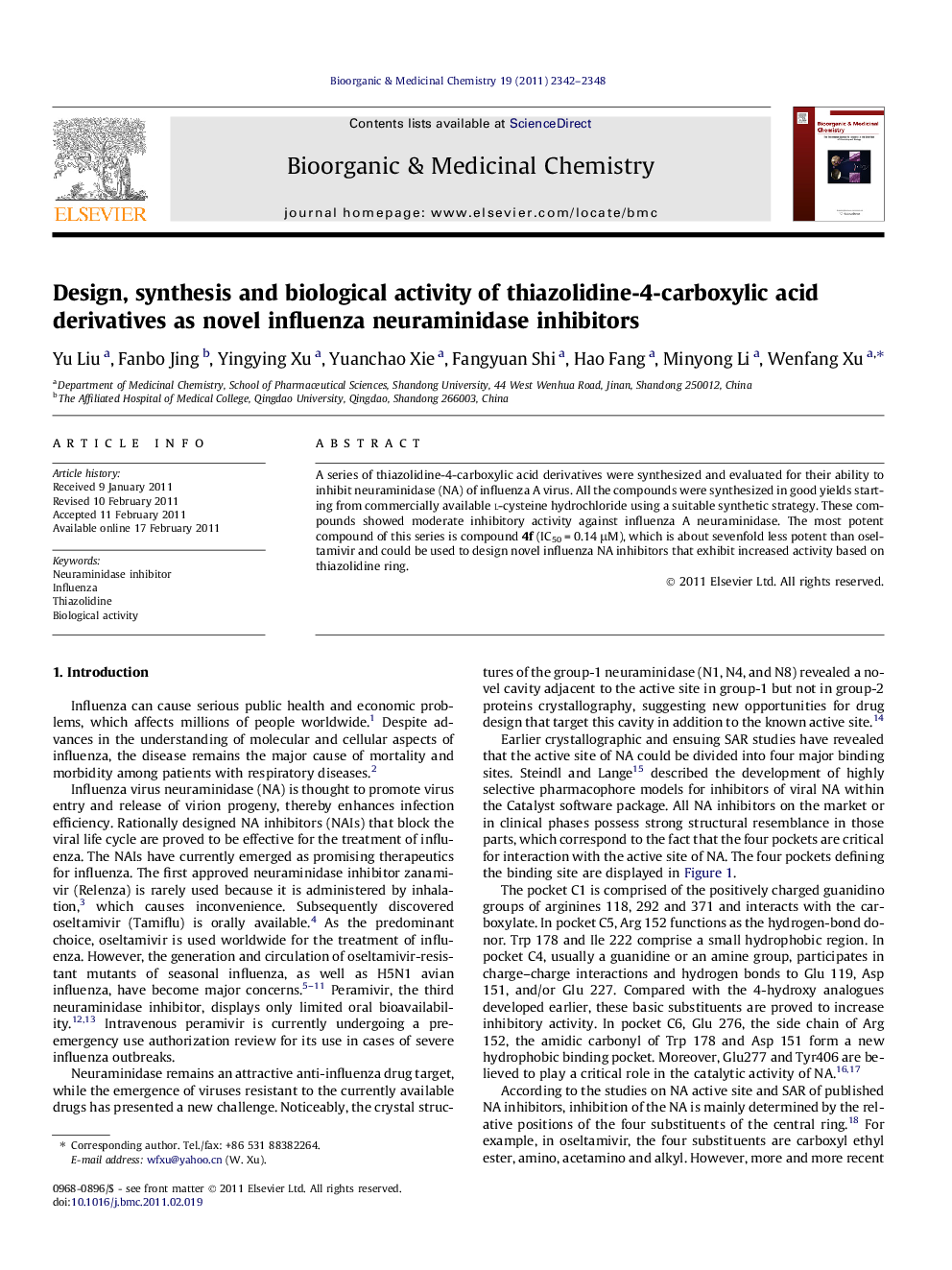
Docking result of compound 4f and oseltamivir (4f is displayed by atom type while oseltamivir is shown in lime line). The active pocket is displayed using Channel pattern (Type: Cavity Depth) with the program MOLCAD Surfaces of Sybyl8.1.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 19, Issue 7, 1 April 2011, Pages 2342–2348