| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1360217 | 981428 | 2009 | 8 صفحه PDF | دانلود رایگان |
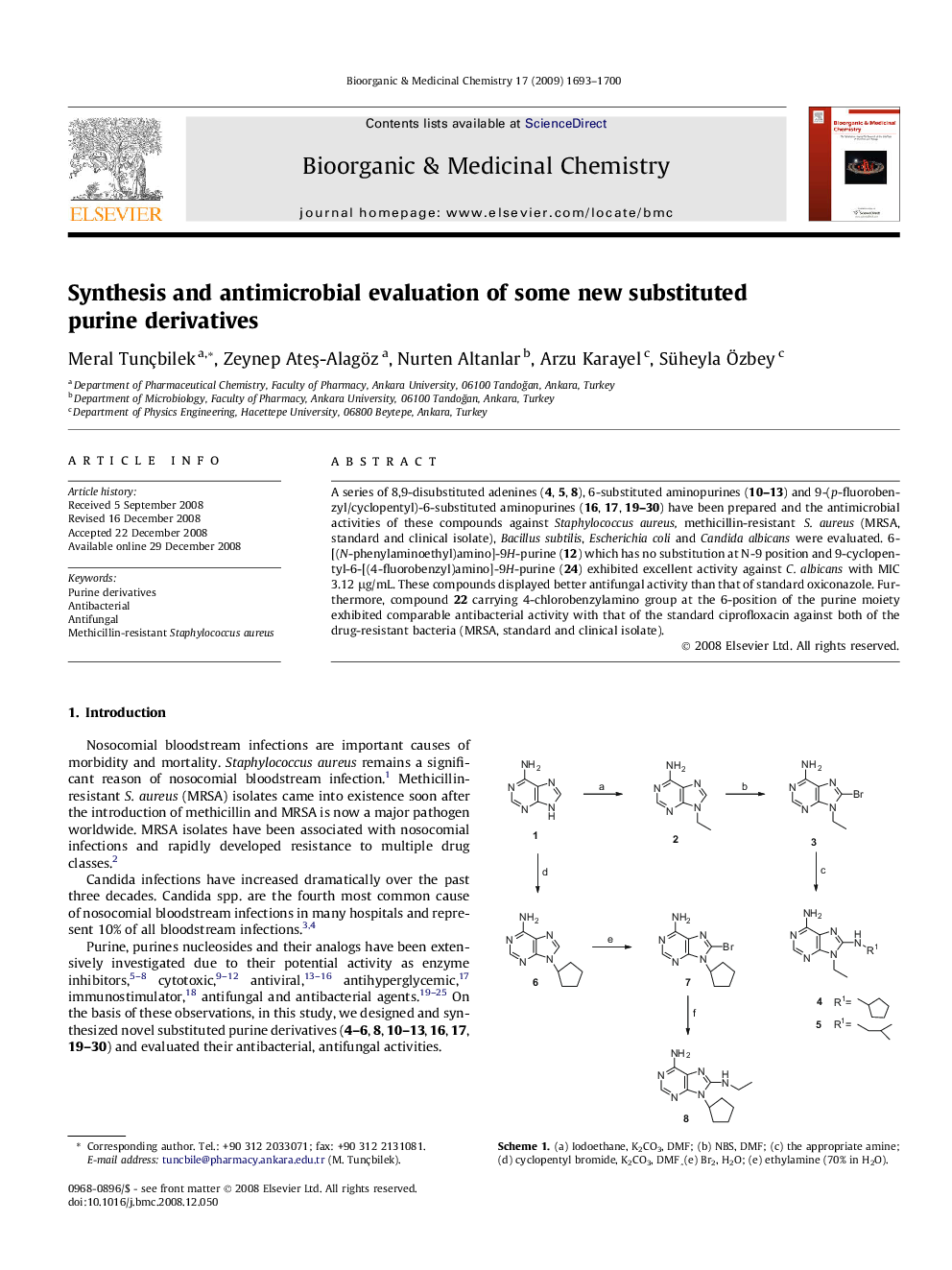
A series of 8,9-disubstituted adenines (4, 5, 8), 6-substituted aminopurines (10–13) and 9-(p-fluorobenzyl/cyclopentyl)-6-substituted aminopurines (16, 17, 19–30) have been prepared and the antimicrobial activities of these compounds against Staphylococcus aureus, methicillin-resistant S. aureus (MRSA, standard and clinical isolate), Bacillus subtilis, Escherichia coli and Candida albicans were evaluated. 6-[(N-phenylaminoethyl)amino]-9H-purine (12) which has no substitution at N-9 position and 9-cyclopentyl-6-[(4-fluorobenzyl)amino]-9H-purine (24) exhibited excellent activity against C. albicans with MIC 3.12 μg/mL. These compounds displayed better antifungal activity than that of standard oxiconazole. Furthermore, compound 22 carrying 4-chlorobenzylamino group at the 6-position of the purine moiety exhibited comparable antibacterial activity with that of the standard ciprofloxacin against both of the drug-resistant bacteria (MRSA, standard and clinical isolate).
Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 17, Issue 4, 15 February 2009, Pages 1693–1700