| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1360287 | 981432 | 2009 | 7 صفحه PDF | دانلود رایگان |
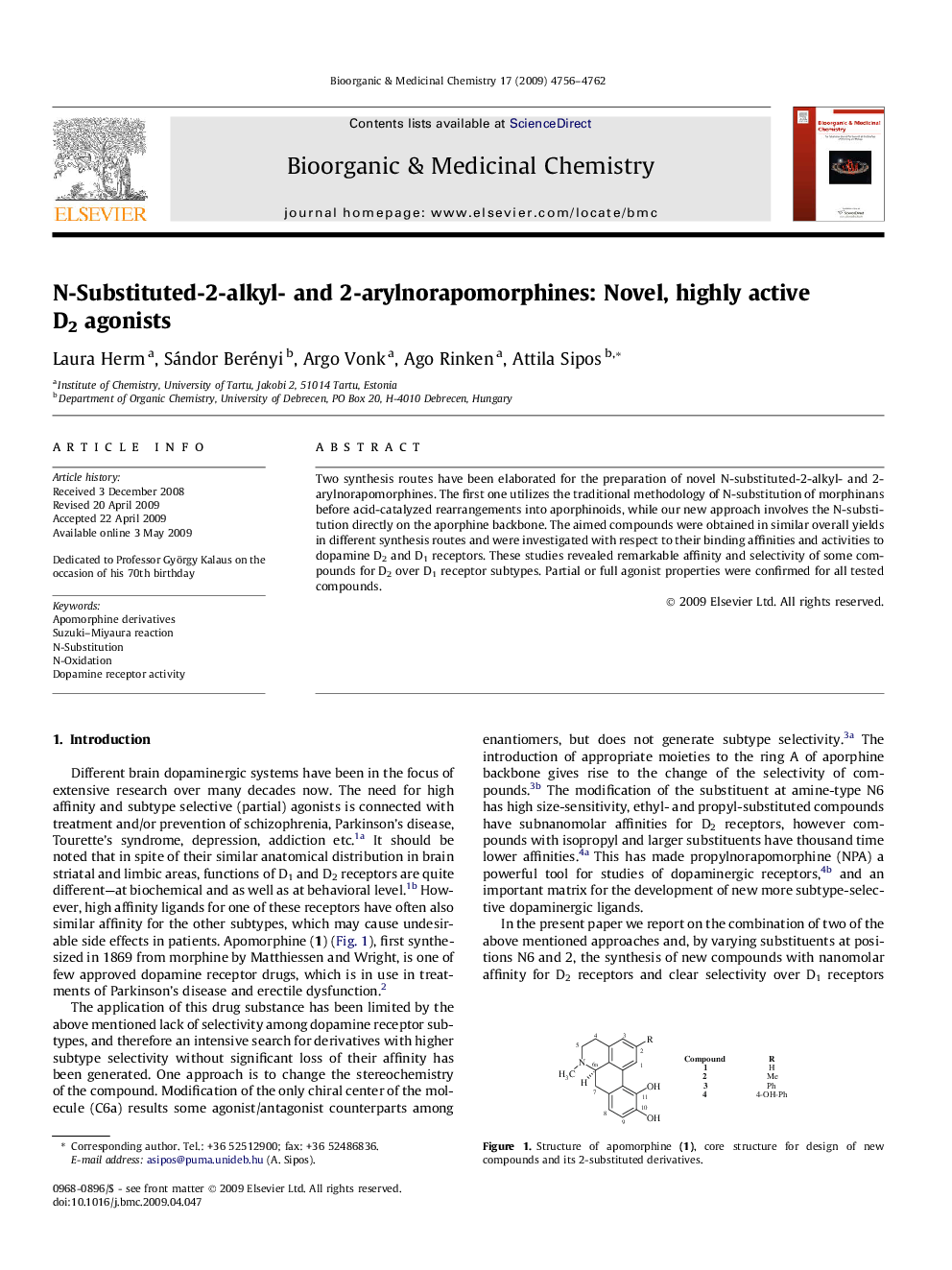
Two synthesis routes have been elaborated for the preparation of novel N-substituted-2-alkyl- and 2-arylnorapomorphines. The first one utilizes the traditional methodology of N-substitution of morphinans before acid-catalyzed rearrangements into aporphinoids, while our new approach involves the N-substitution directly on the aporphine backbone. The aimed compounds were obtained in similar overall yields in different synthesis routes and were investigated with respect to their binding affinities and activities to dopamine D2 and D1 receptors. These studies revealed remarkable affinity and selectivity of some compounds for D2 over D1 receptor subtypes. Partial or full agonist properties were confirmed for all tested compounds.
Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 17, Issue 13, 1 July 2009, Pages 4756–4762