| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1361581 | 981467 | 2007 | 8 صفحه PDF | دانلود رایگان |
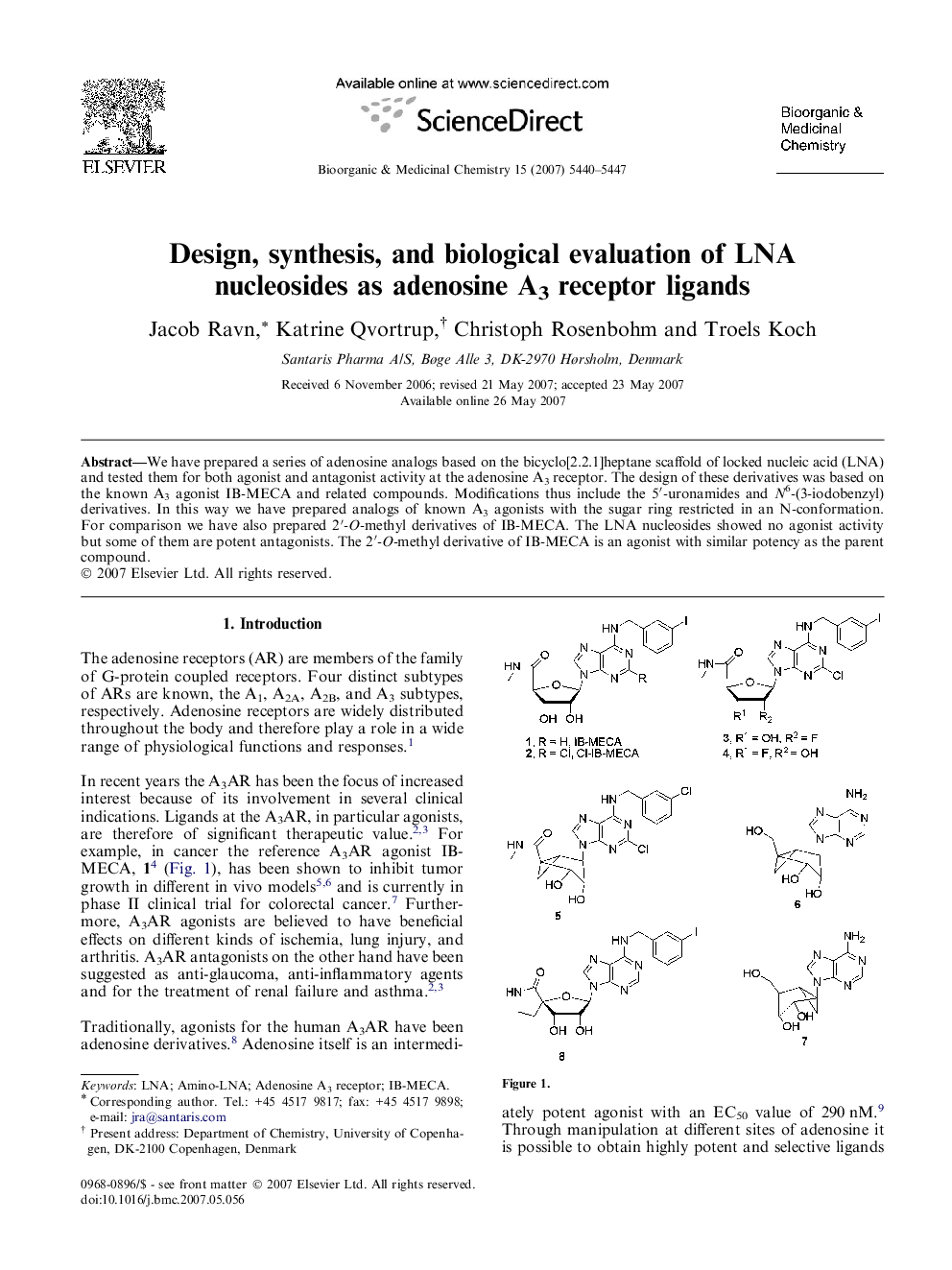
We have prepared a series of adenosine analogs based on the bicyclo[2.2.1]heptane scaffold of locked nucleic acid (LNA) and tested them for both agonist and antagonist activity at the adenosine A3 receptor. The design of these derivatives was based on the known A3 agonist IB-MECA and related compounds. Modifications thus include the 5′-uronamides and N6-(3-iodobenzyl) derivatives. In this way we have prepared analogs of known A3 agonists with the sugar ring restricted in an N-conformation. For comparison we have also prepared 2′-O-methyl derivatives of IB-MECA. The LNA nucleosides showed no agonist activity but some of them are potent antagonists. The 2′-O-methyl derivative of IB-MECA is an agonist with similar potency as the parent compound.
Locked nucleic acid (LNA) analogs of the A3AR agonist IB-MECA.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 15, Issue 16, 15 August 2007, Pages 5440–5447