| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1363146 | 981504 | 2009 | 5 صفحه PDF | دانلود رایگان |
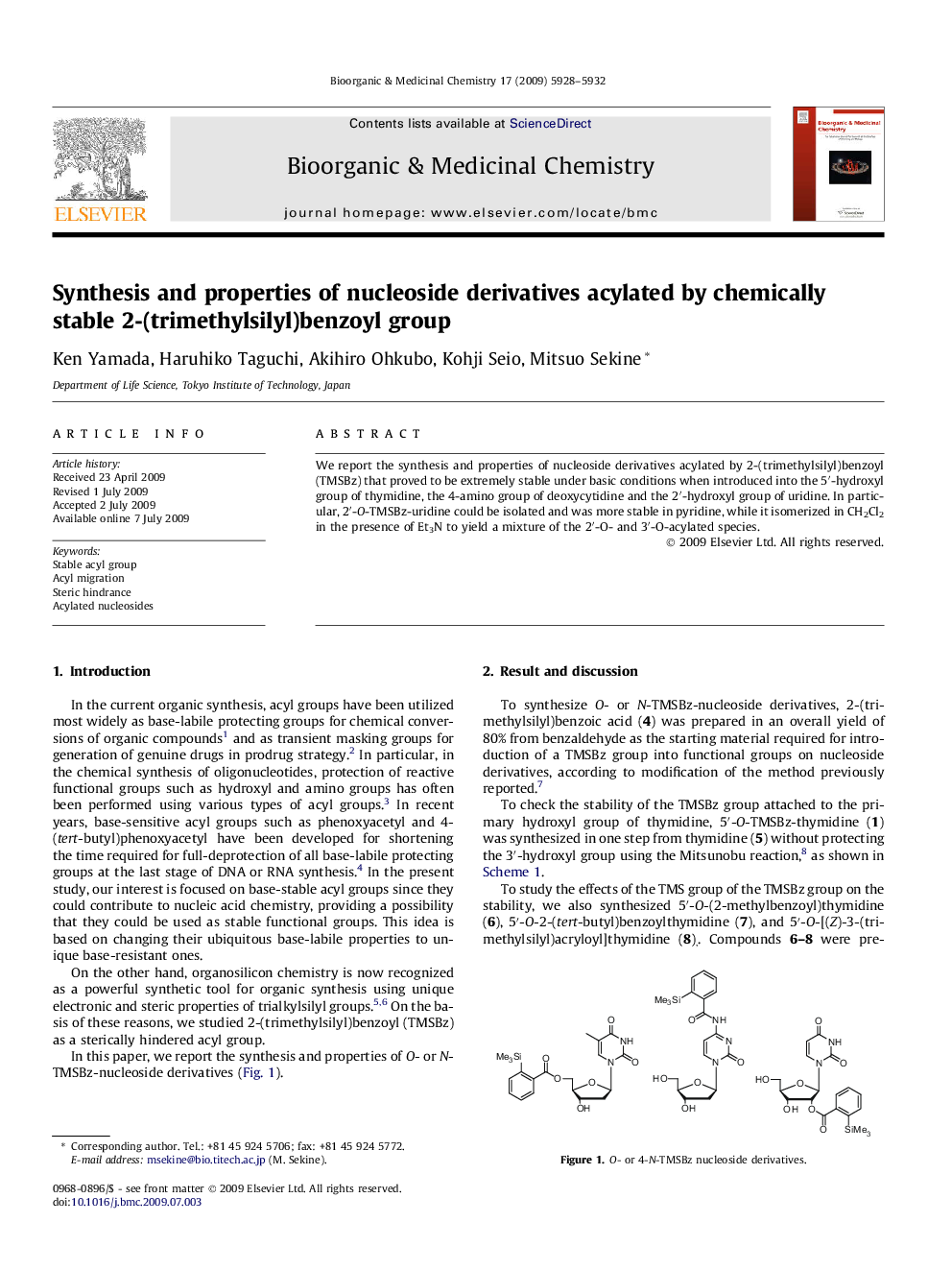
We report the synthesis and properties of nucleoside derivatives acylated by 2-(trimethylsilyl)benzoyl (TMSBz) that proved to be extremely stable under basic conditions when introduced into the 5′-hydroxyl group of thymidine, the 4-amino group of deoxycytidine and the 2′-hydroxyl group of uridine. In particular, 2′-O-TMSBz-uridine could be isolated and was more stable in pyridine, while it isomerized in CH2Cl2 in the presence of Et3N to yield a mixture of the 2′-O- and 3′-O-acylated species.
We developed new chemically stable 2-(trimethylsilyl)benzoyl (TMSBz) group. Nucleoside derivatives acylated by TMSBz group was synthesized and analyzed its chemical stability under basic conditions. As a result, TMSBz group showed very high stability comparing with 2-methylbenzoyl and (Z)-3-(trimethylsilyl)acryloyl group.Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 17, Issue 16, 15 August 2009, Pages 5928–5932