| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 146486 | 456371 | 2015 | 9 صفحه PDF | دانلود رایگان |
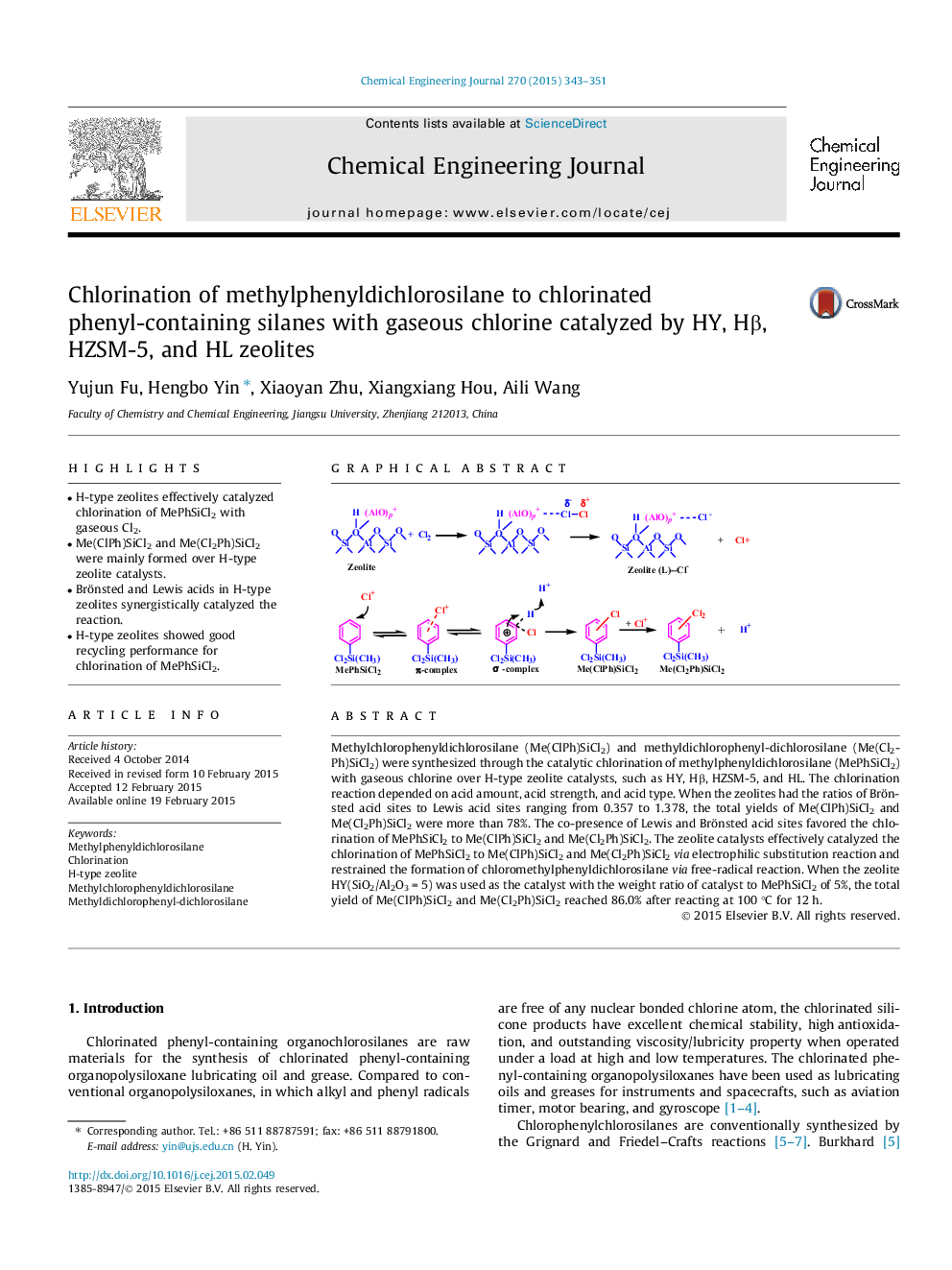
• H-type zeolites effectively catalyzed chlorination of MePhSiCl2 with gaseous Cl2.
• Me(ClPh)SiCl2 and Me(Cl2Ph)SiCl2 were mainly formed over H-type zeolite catalysts.
• Brönsted and Lewis acids in H-type zeolites synergistically catalyzed the reaction.
• H-type zeolites showed good recycling performance for chlorination of MePhSiCl2.
Methylchlorophenyldichlorosilane (Me(ClPh)SiCl2) and methyldichlorophenyl-dichlorosilane (Me(Cl2Ph)SiCl2) were synthesized through the catalytic chlorination of methylphenyldichlorosilane (MePhSiCl2) with gaseous chlorine over H-type zeolite catalysts, such as HY, Hβ, HZSM-5, and HL. The chlorination reaction depended on acid amount, acid strength, and acid type. When the zeolites had the ratios of Brönsted acid sites to Lewis acid sites ranging from 0.357 to 1.378, the total yields of Me(ClPh)SiCl2 and Me(Cl2Ph)SiCl2 were more than 78%. The co-presence of Lewis and Brönsted acid sites favored the chlorination of MePhSiCl2 to Me(ClPh)SiCl2 and Me(Cl2Ph)SiCl2. The zeolite catalysts effectively catalyzed the chlorination of MePhSiCl2 to Me(ClPh)SiCl2 and Me(Cl2Ph)SiCl2via electrophilic substitution reaction and restrained the formation of chloromethylphenyldichlorosilane via free-radical reaction. When the zeolite HY(SiO2/Al2O3 = 5) was used as the catalyst with the weight ratio of catalyst to MePhSiCl2 of 5%, the total yield of Me(ClPh)SiCl2 and Me(Cl2Ph)SiCl2 reached 86.0% after reacting at 100 °C for 12 h.
Figure optionsDownload as PowerPoint slide
Journal: Chemical Engineering Journal - Volume 270, 15 June 2015, Pages 343–351