| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 146579 | 456373 | 2015 | 8 صفحه PDF | دانلود رایگان |
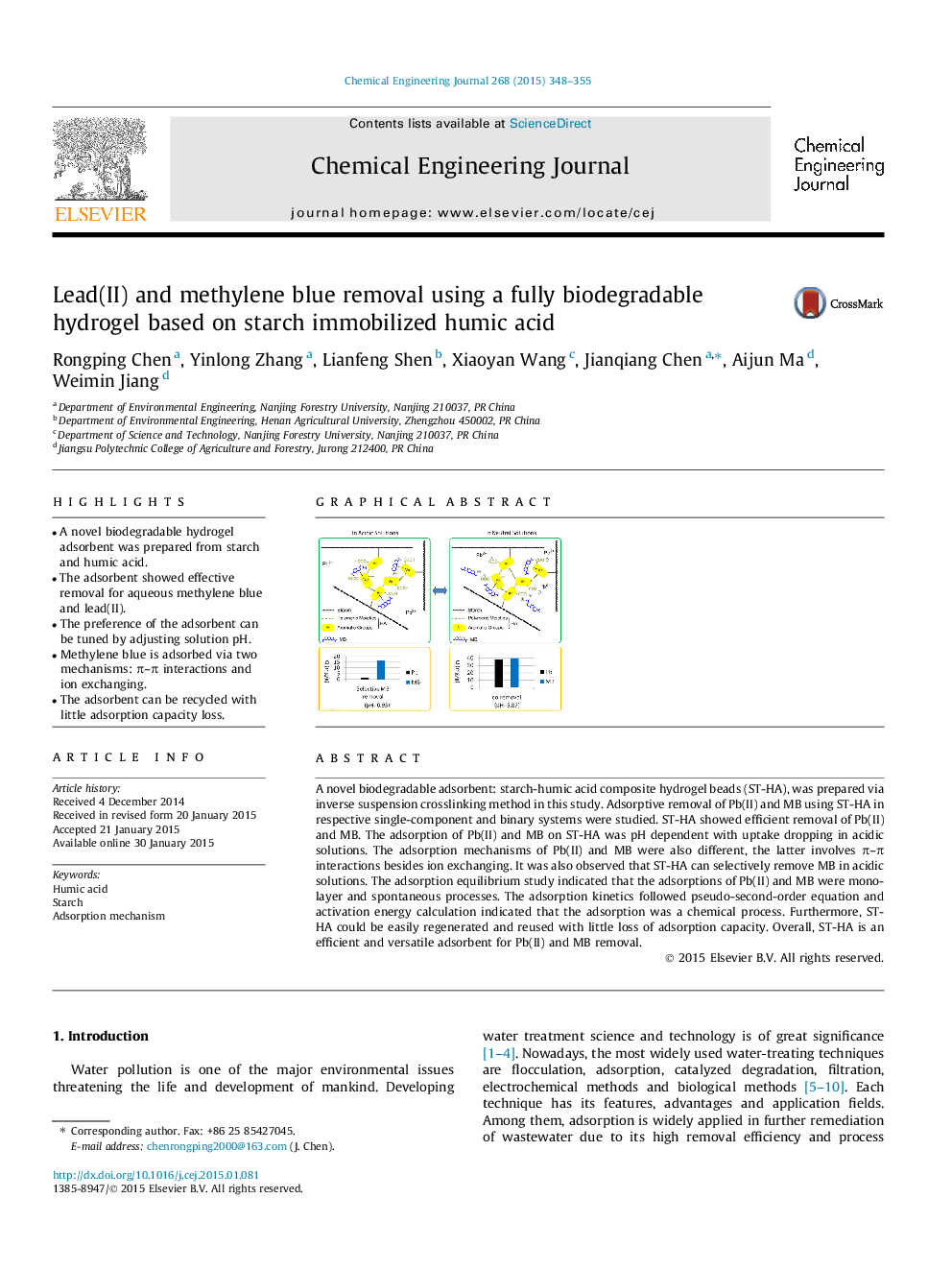
• A novel biodegradable hydrogel adsorbent was prepared from starch and humic acid.
• The adsorbent showed effective removal for aqueous methylene blue and lead(II).
• The preference of the adsorbent can be tuned by adjusting solution pH.
• Methylene blue is adsorbed via two mechanisms: π–π interactions and ion exchanging.
• The adsorbent can be recycled with little adsorption capacity loss.
A novel biodegradable adsorbent: starch-humic acid composite hydrogel beads (ST-HA), was prepared via inverse suspension crosslinking method in this study. Adsorptive removal of Pb(II) and MB using ST-HA in respective single-component and binary systems were studied. ST-HA showed efficient removal of Pb(II) and MB. The adsorption of Pb(II) and MB on ST-HA was pH dependent with uptake dropping in acidic solutions. The adsorption mechanisms of Pb(II) and MB were also different, the latter involves π–π interactions besides ion exchanging. It was also observed that ST-HA can selectively remove MB in acidic solutions. The adsorption equilibrium study indicated that the adsorptions of Pb(II) and MB were monolayer and spontaneous processes. The adsorption kinetics followed pseudo-second-order equation and activation energy calculation indicated that the adsorption was a chemical process. Furthermore, ST-HA could be easily regenerated and reused with little loss of adsorption capacity. Overall, ST-HA is an efficient and versatile adsorbent for Pb(II) and MB removal.
Figure optionsDownload as PowerPoint slide
Journal: Chemical Engineering Journal - Volume 268, 15 May 2015, Pages 348–355