| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 147985 | 456403 | 2014 | 12 صفحه PDF | دانلود رایگان |
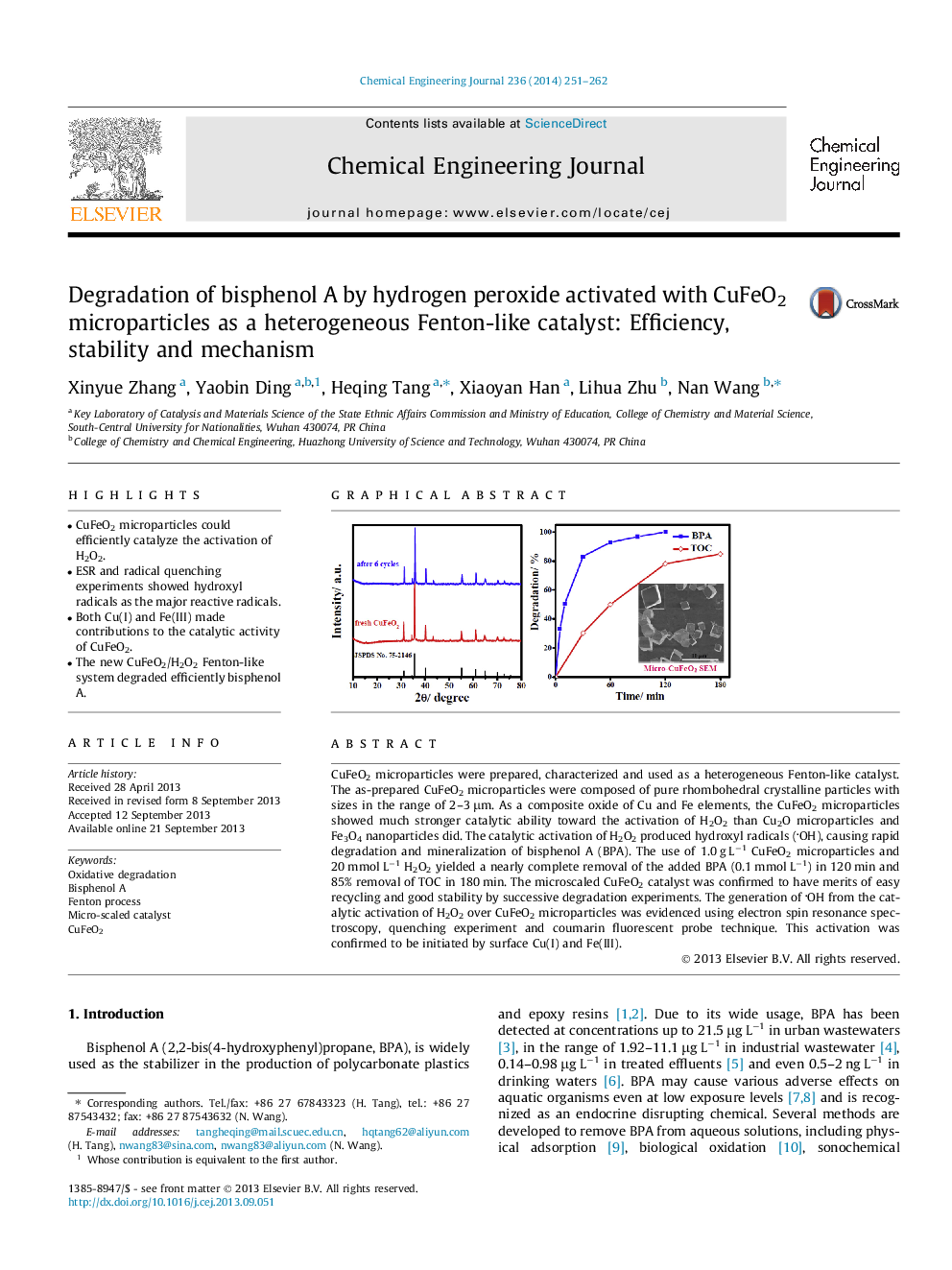
• CuFeO2 microparticles could efficiently catalyze the activation of H2O2.
• ESR and radical quenching experiments showed hydroxyl radicals as the major reactive radicals.
• Both Cu(I) and Fe(III) made contributions to the catalytic activity of CuFeO2.
• The new CuFeO2/H2O2 Fenton-like system degraded efficiently bisphenol A.
CuFeO2 microparticles were prepared, characterized and used as a heterogeneous Fenton-like catalyst. The as-prepared CuFeO2 microparticles were composed of pure rhombohedral crystalline particles with sizes in the range of 2–3 μm. As a composite oxide of Cu and Fe elements, the CuFeO2 microparticles showed much stronger catalytic ability toward the activation of H2O2 than Cu2O microparticles and Fe3O4 nanoparticles did. The catalytic activation of H2O2 produced hydroxyl radicals (OH), causing rapid degradation and mineralization of bisphenol A (BPA). The use of 1.0 g L−1 CuFeO2 microparticles and 20 mmol L−1 H2O2 yielded a nearly complete removal of the added BPA (0.1 mmol L−1) in 120 min and 85% removal of TOC in 180 min. The microscaled CuFeO2 catalyst was confirmed to have merits of easy recycling and good stability by successive degradation experiments. The generation of OH from the catalytic activation of H2O2 over CuFeO2 microparticles was evidenced using electron spin resonance spectroscopy, quenching experiment and coumarin fluorescent probe technique. This activation was confirmed to be initiated by surface Cu(I) and Fe(III).
Figure optionsDownload as PowerPoint slide
Journal: Chemical Engineering Journal - Volume 236, 15 January 2014, Pages 251–262