| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 2029622 | 1070930 | 2016 | 11 صفحه PDF | دانلود رایگان |
• Mutations in cMyBP-C linked to familial HCM have distinct structural consequences
• Mutations impeding domain folding are linked to late-onset severe hypertrophy
• A mutation linked to early-onset HCM may only interfere with protein interactions
• Bioinformatics and modeling largely predicted the experimentally observed results
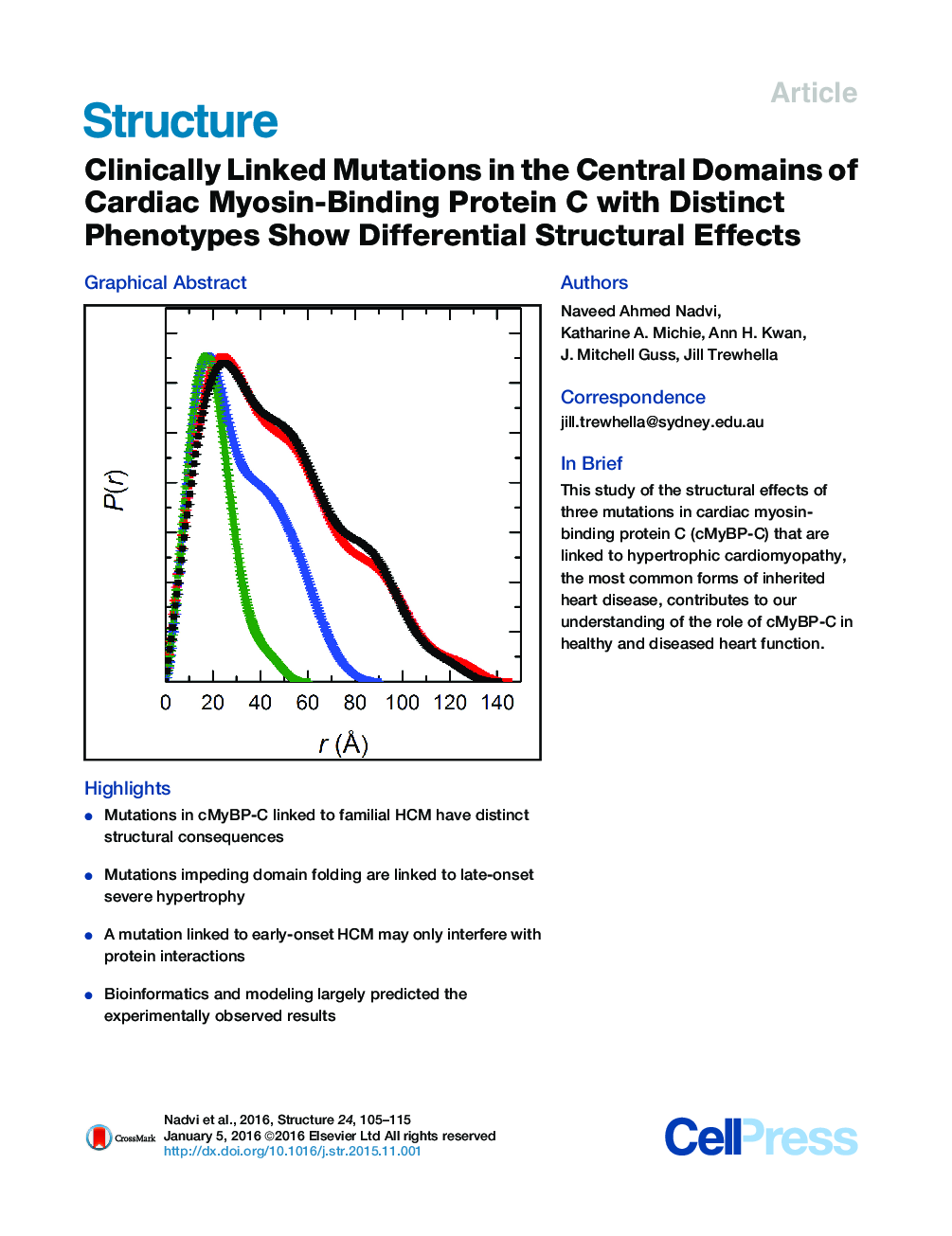
SummaryThe structural effects of three missense mutations clinically linked to hypertrophic cardiomyopathy (HCM) and located in the central domains of cardiac myosin-binding protein C (cMyBP-C) have been determined using small-angle scattering, infrared spectroscopy, and nuclear magnetic resonance spectroscopy. Bioinformatics and modeling were used to initially predict the expected structural impacts and assess the broader implications for function based on sequence conservation patterns. The experimental results generally affirm the predictions that two of the mutations (D745G, P873H) disrupt domain folding, while the third (R820Q) is likely to be entirely solvent exposed and thus more likely to have its impact through its interactions within the sarcomere. Each of the mutations is associated with distinct disease phenotypes, with respect to severity, stage of onset, and end phase. The results are discussed in terms of understanding key structural features of these domains essential for healthy function and the role they may play in disease development.
Graphical AbstractFigure optionsDownload high-quality image (164 K)Download as PowerPoint slide
Journal: - Volume 24, Issue 1, 5 January 2016, Pages 105–115