| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 205997 | 461135 | 2014 | 7 صفحه PDF | دانلود رایگان |
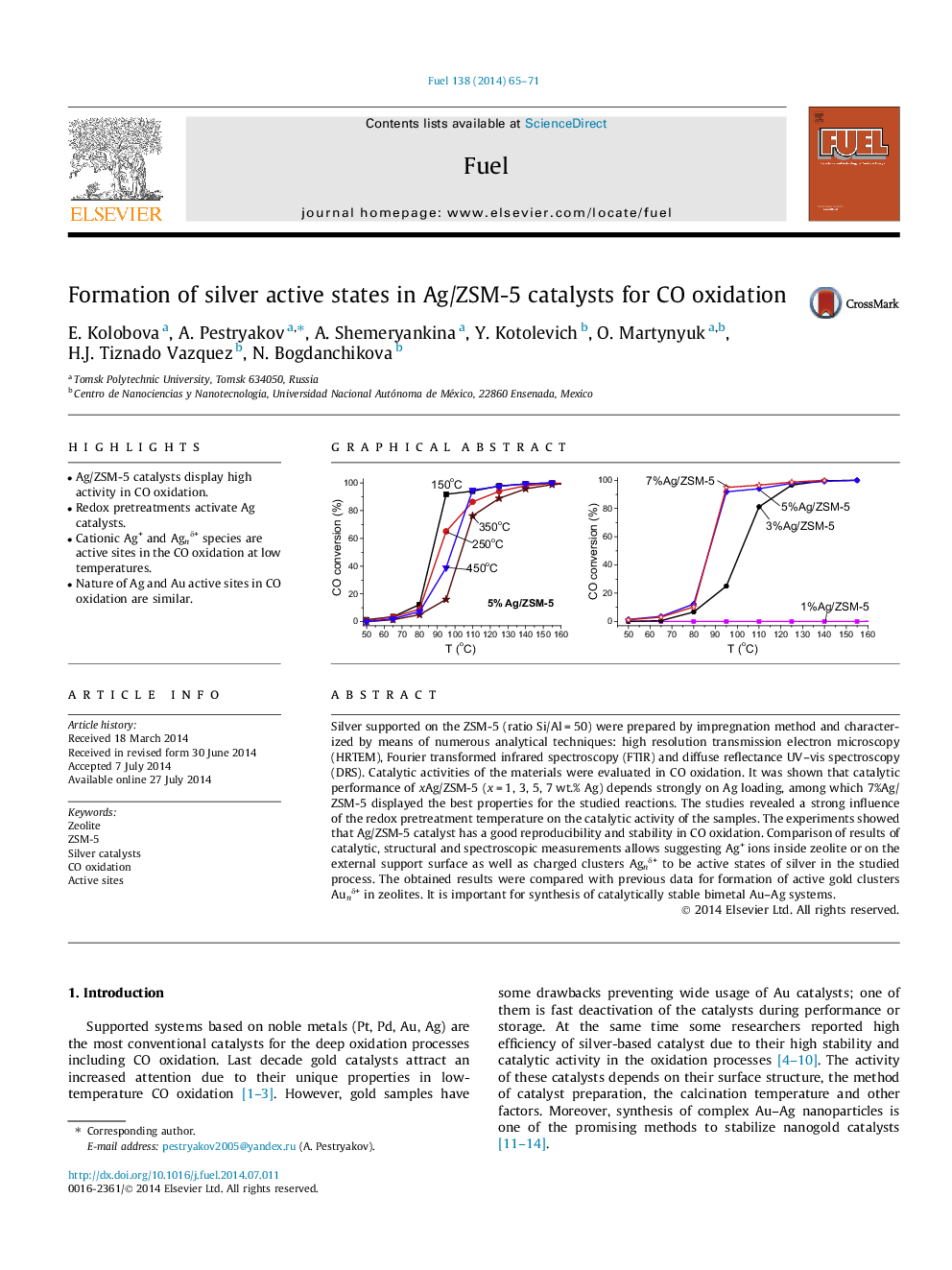
• Ag/ZSM-5 catalysts display high activity in CO oxidation.
• Redox pretreatments activate Ag catalysts.
• Cationic Ag+ and Agnδ+ species are active sites in the CO oxidation at low temperatures.
• Nature of Ag and Au active sites in CO oxidation are similar.
Silver supported on the ZSM-5 (ratio Si/Al = 50) were prepared by impregnation method and characterized by means of numerous analytical techniques: high resolution transmission electron microscopy (HRTEM), Fourier transformed infrared spectroscopy (FTIR) and diffuse reflectance UV–vis spectroscopy (DRS). Catalytic activities of the materials were evaluated in CO oxidation. It was shown that catalytic performance of xAg/ZSM-5 (x = 1, 3, 5, 7 wt.% Ag) depends strongly on Ag loading, among which 7%Ag/ZSM-5 displayed the best properties for the studied reactions. The studies revealed a strong influence of the redox pretreatment temperature on the catalytic activity of the samples. The experiments showed that Ag/ZSM-5 catalyst has a good reproducibility and stability in CO oxidation. Comparison of results of catalytic, structural and spectroscopic measurements allows suggesting Ag+ ions inside zeolite or on the external support surface as well as charged clusters Agnδ+ to be active states of silver in the studied process. The obtained results were compared with previous data for formation of active gold clusters Aunδ+ in zeolites. It is important for synthesis of catalytically stable bimetal Au–Ag systems.
Figure optionsDownload as PowerPoint slide
Journal: Fuel - Volume 138, 15 December 2014, Pages 65–71