| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 5277386 | 1385579 | 2008 | 4 صفحه PDF | دانلود رایگان |
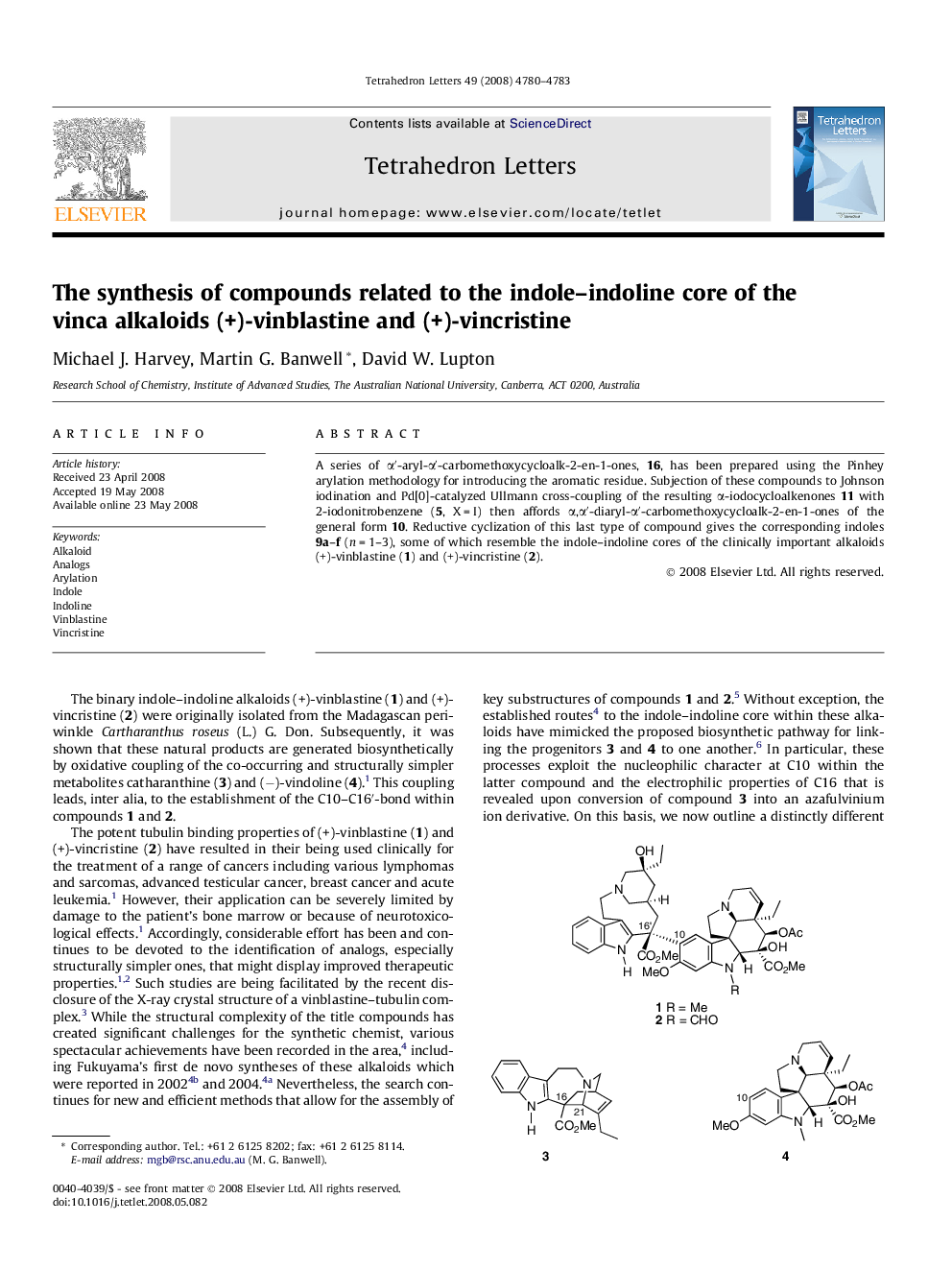
A series of αâ²-aryl-αâ²-carbomethoxycycloalk-2-en-1-ones, 16, has been prepared using the Pinhey arylation methodology for introducing the aromatic residue. Subjection of these compounds to Johnson iodination and Pd[0]-catalyzed Ullmann cross-coupling of the resulting α-iodocycloalkenones 11 with 2-iodonitrobenzene (5, X = I) then affords α,αâ²-diaryl-αâ²-carbomethoxycycloalk-2-en-1-ones of the general form 10. Reductive cyclization of this last type of compound gives the corresponding indoles 9a-f (n = 1-3), some of which resemble the indole-indoline cores of the clinically important alkaloids (+)-vinblastine (1) and (+)-vincristine (2).
Analogs, 9, of the indole-indoline core of the title alkaloids have been prepared, via intermediates 10, from α-iodoenones of the general form 11.
Journal: Tetrahedron Letters - Volume 49, Issue 32, 4 August 2008, Pages 4780-4783