| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 5280424 | 1385647 | 2008 | 4 صفحه PDF | دانلود رایگان |
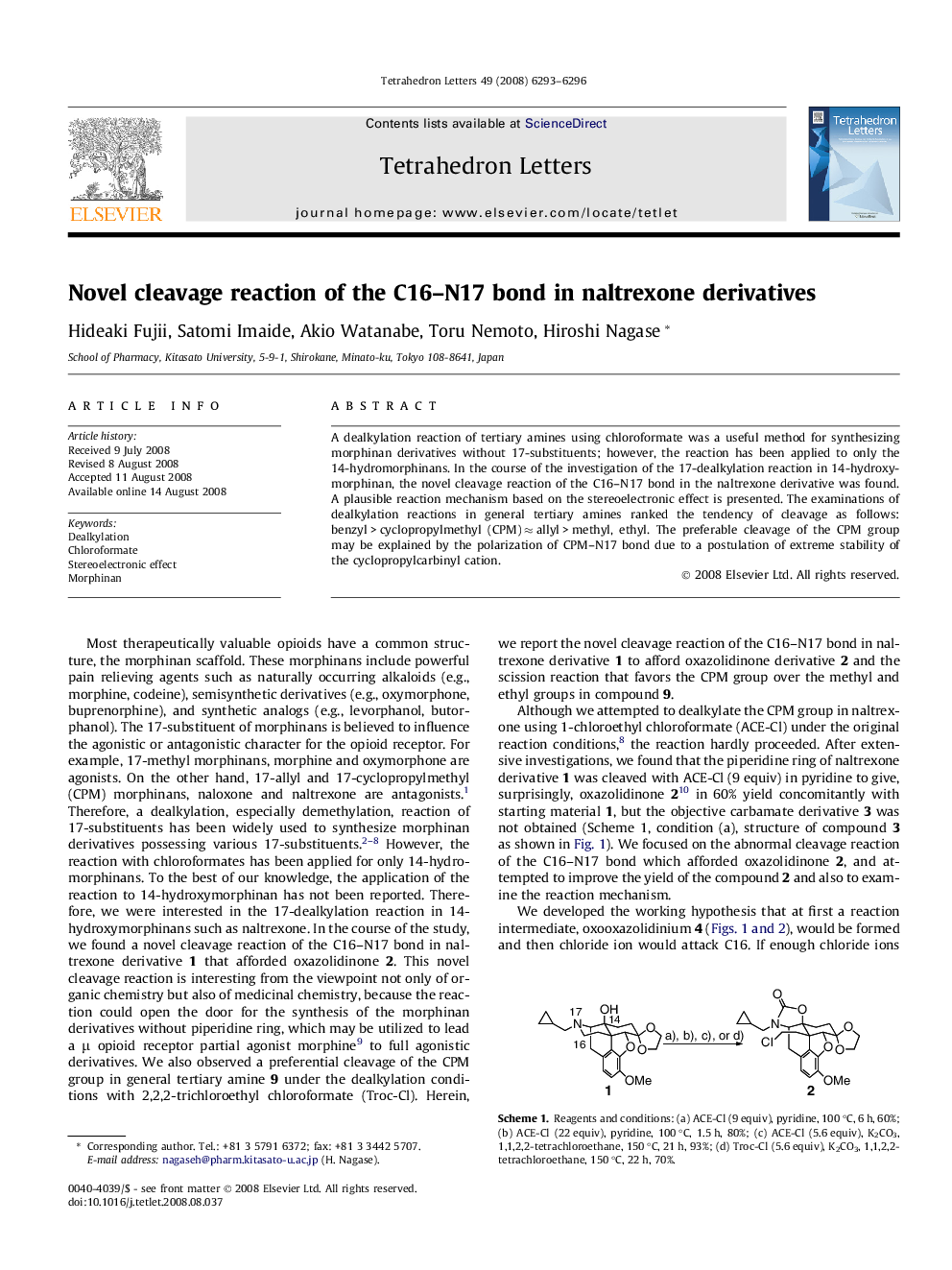
A dealkylation reaction of tertiary amines using chloroformate was a useful method for synthesizing morphinan derivatives without 17-substituents; however, the reaction has been applied to only the 14-hydromorphinans. In the course of the investigation of the 17-dealkylation reaction in 14-hydroxymorphinan, the novel cleavage reaction of the C16-N17 bond in the naltrexone derivative was found. A plausible reaction mechanism based on the stereoelectronic effect is presented. The examinations of dealkylation reactions in general tertiary amines ranked the tendency of cleavage as follows: benzyl > cyclopropylmethyl (CPM) â allyl > methyl, ethyl. The preferable cleavage of the CPM group may be explained by the polarization of CPM-N17 bond due to a postulation of extreme stability of the cyclopropylcarbinyl cation.
The treatment of 14-OH morphinans with 2,2,2-trichloroethyl chloroformate (Troc-Cl) cleaved the C16-N17 bond of the morphinan derivative to afford a novel oxazolidinone derivative.
Journal: Tetrahedron Letters - Volume 49, Issue 44, 27 October 2008, Pages 6293-6296