| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 65312 | 48388 | 2014 | 9 صفحه PDF | دانلود رایگان |
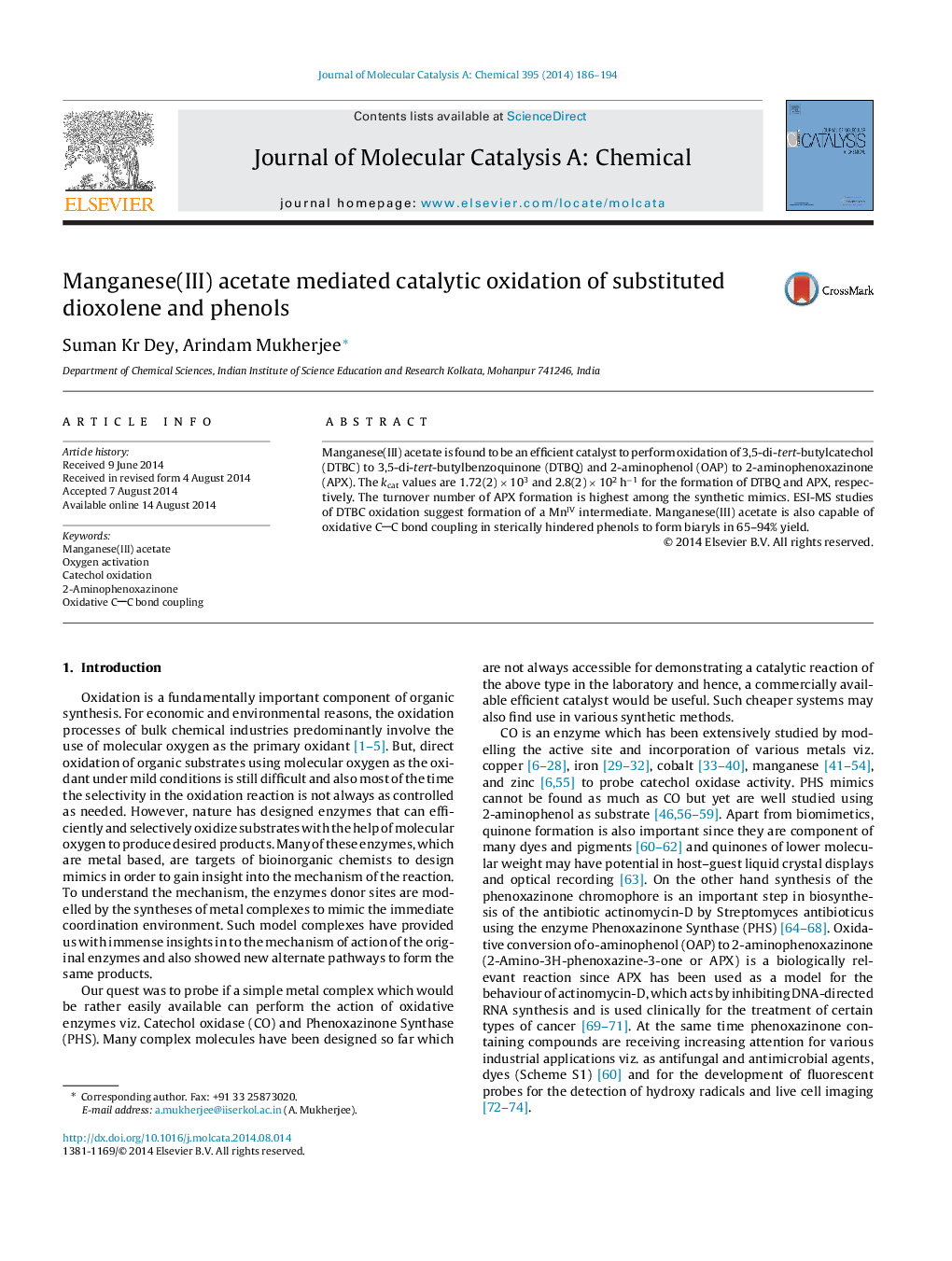
• MnIII acetate catalytically oxidizes 3,5-di-tert-butylcatechol to quinone with high turnover.
• Oxidation of DTBC to DTBQ goes through a MnIV-semiquinone species as per ESI-MS data.
• MnIII acetate also efficiently catalyzes oxidation of 2-aminophenol to 2-aminophenoxazinone.
• MnIII acetate oxidatively couples sterically hindered phenols to yield biaryls.
Manganese(III) acetate is found to be an efficient catalyst to perform oxidation of 3,5-di-tert-butylcatechol (DTBC) to 3,5-di-tert-butylbenzoquinone (DTBQ) and 2-aminophenol (OAP) to 2-aminophenoxazinone (APX). The kcat values are 1.72(2) × 103 and 2.8(2) × 102 h−1 for the formation of DTBQ and APX, respectively. The turnover number of APX formation is highest among the synthetic mimics. ESI-MS studies of DTBC oxidation suggest formation of a MnIV intermediate. Manganese(III) acetate is also capable of oxidative CC bond coupling in sterically hindered phenols to form biaryls in 65–94% yield.
Figure optionsDownload high-quality image (96 K)Download as PowerPoint slide
Journal: Journal of Molecular Catalysis A: Chemical - Volume 395, December 2014, Pages 186–194