| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1231524 | 1495259 | 2013 | 11 صفحه PDF | دانلود رایگان |
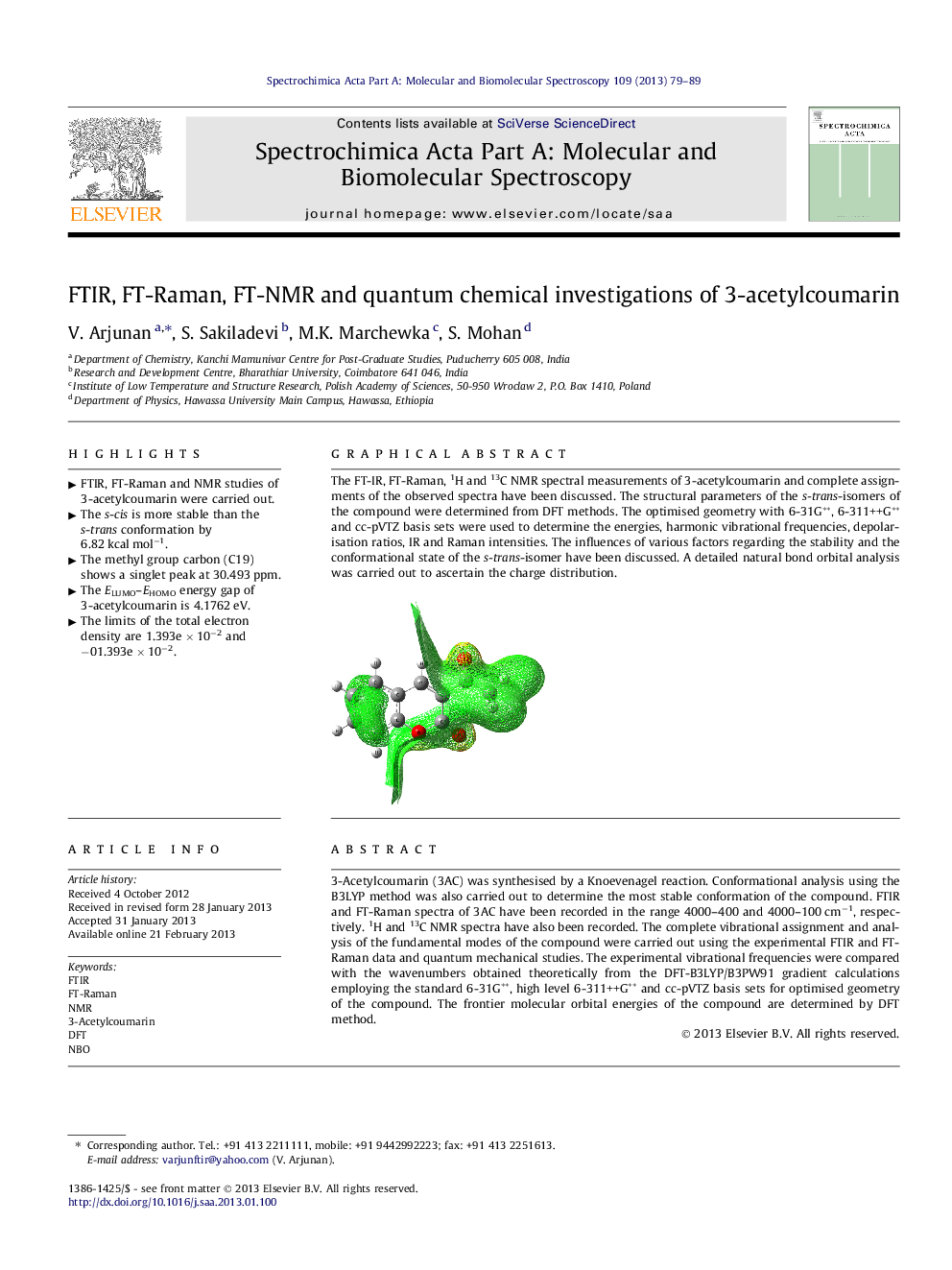
3-Acetylcoumarin (3AC) was synthesised by a Knoevenagel reaction. Conformational analysis using the B3LYP method was also carried out to determine the most stable conformation of the compound. FTIR and FT-Raman spectra of 3AC have been recorded in the range 4000–400 and 4000–100 cm−1, respectively. 1H and 13C NMR spectra have also been recorded. The complete vibrational assignment and analysis of the fundamental modes of the compound were carried out using the experimental FTIR and FT-Raman data and quantum mechanical studies. The experimental vibrational frequencies were compared with the wavenumbers obtained theoretically from the DFT-B3LYP/B3PW91 gradient calculations employing the standard 6-31G**, high level 6-311++G** and cc-pVTZ basis sets for optimised geometry of the compound. The frontier molecular orbital energies of the compound are determined by DFT method.
The FT-IR, FT-Raman, 1H and 13C NMR spectral measurements of 3-acetylcoumarin and complete assignments of the observed spectra have been discussed. The structural parameters of the s-trans-isomers of the compound were determined from DFT methods. The optimised geometry with 6-31G**, 6-311++G** and cc-pVTZ basis sets were used to determine the energies, harmonic vibrational frequencies, depolarisation ratios, IR and Raman intensities. The influences of various factors regarding the stability and the conformational state of the s-trans-isomer have been discussed. A detailed natural bond orbital analysis was carried out to ascertain the charge distribution.Figure optionsDownload as PowerPoint slideHighlights
► FTIR, FT-Raman and NMR studies of 3-acetylcoumarin were carried out.
► The s-cis is more stable than the s-trans conformation by 6.82 kcal mol−1.
► The methyl group carbon (C19) shows a singlet peak at 30.493 ppm.
► The ELUMO–EHOMO energy gap of 3-acetylcoumarin is 4.1762 eV.
► The limits of the total electron density are 1.393e × 10−2 and −01.393e × 10−2.
Journal: Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy - Volume 109, 15 May 2013, Pages 79–89