| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1271449 | 1497453 | 2015 | 6 صفحه PDF | دانلود رایگان |
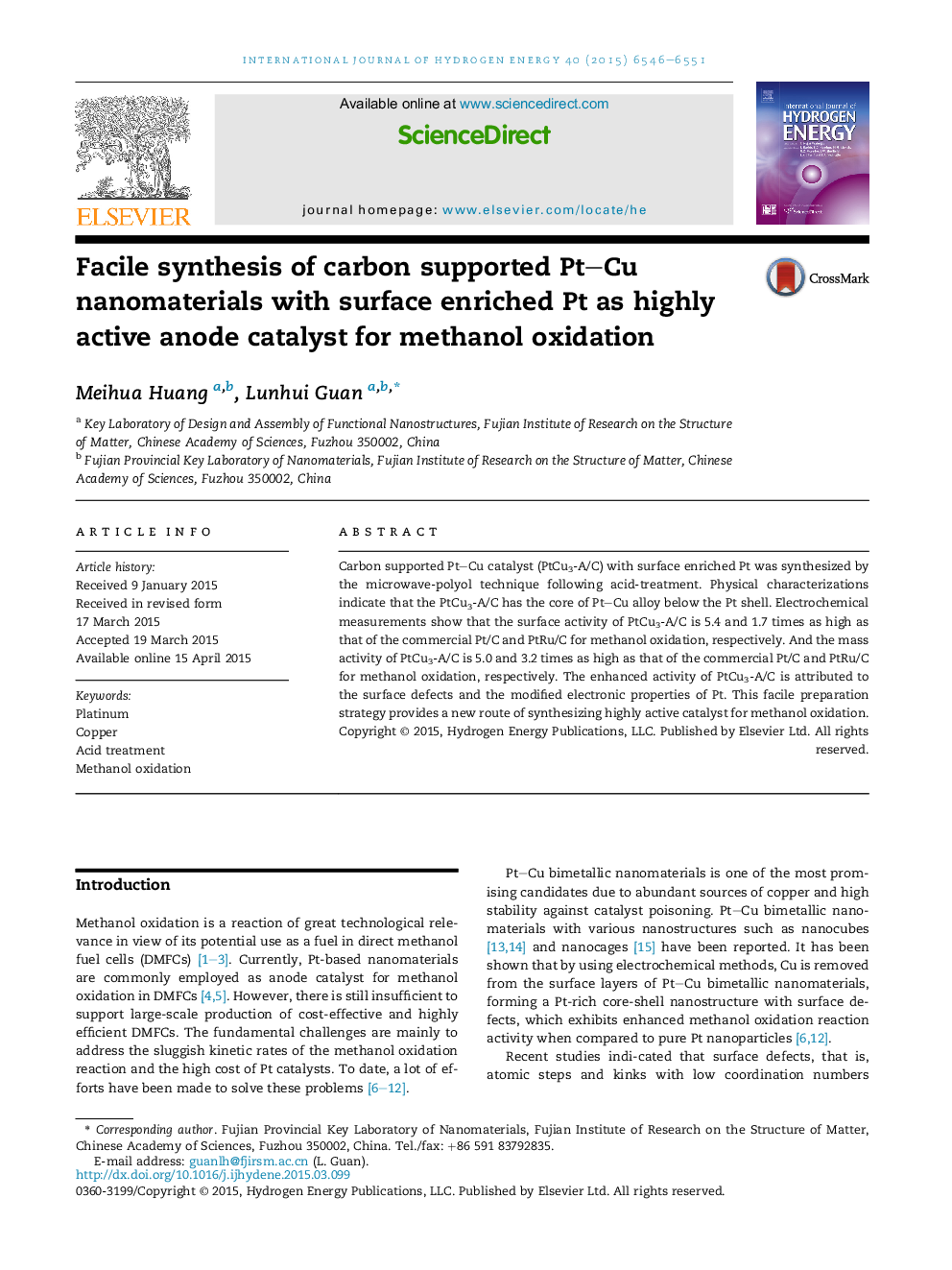
• The PtCu3-A/C with a Pt–Cu alloy core and surface Pt defects was synthesized.
• The PtCu3-A/C has higher mass and surface activity for methanol oxidation.
• The chemical dealloying is suitable to prepare highly active Pt-based catalysts.
Carbon supported Pt–Cu catalyst (PtCu3-A/C) with surface enriched Pt was synthesized by the microwave-polyol technique following acid-treatment. Physical characterizations indicate that the PtCu3-A/C has the core of Pt–Cu alloy below the Pt shell. Electrochemical measurements show that the surface activity of PtCu3-A/C is 5.4 and 1.7 times as high as that of the commercial Pt/C and PtRu/C for methanol oxidation, respectively. And the mass activity of PtCu3-A/C is 5.0 and 3.2 times as high as that of the commercial Pt/C and PtRu/C for methanol oxidation, respectively. The enhanced activity of PtCu3-A/C is attributed to the surface defects and the modified electronic properties of Pt. This facile preparation strategy provides a new route of synthesizing highly active catalyst for methanol oxidation.
The PtCu3-A/C contained a Pt–Cu core and Pt shell with surface defects. The PtCu3-A/C has mass activity and surface activity 5.0 and 5.4 times higher than those of Pt/C for methanol oxidation. And the mass activity and surface activity of PtCu3-A/C are 3.2 and 1.7 times higher than those of PtRu/C for methanol oxidation. The enhanced activity of PtCu3-A/C is attributed to the surface defects and the modified electronic properties of Pt. This facile preparation strategy provides a new route of synthesizing highly active catalyst for methanol oxidation.Figure optionsDownload as PowerPoint slide
Journal: International Journal of Hydrogen Energy - Volume 40, Issue 20, 1 June 2015, Pages 6546–6551